Energy Metabolism Is Altered in Radioresistant Rectal Cancer
- PMID: 37108244
- PMCID: PMC10138551
- DOI: 10.3390/ijms24087082
Energy Metabolism Is Altered in Radioresistant Rectal Cancer
Abstract
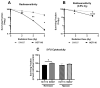
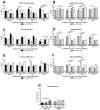
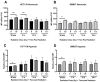
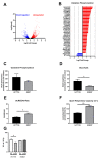
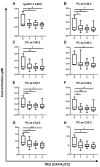
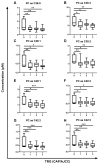
Resistance to neoadjuvant chemoradiation therapy is a significant clinical challenge in the management of rectal cancer. There is an unmet need to identify the underlying mechanisms of treatment resistance to enable the development of biomarkers predictive of response and novel treatment strategies to improve therapeutic response. In this study, an in vitro model of inherently radioresistant rectal cancer was identified and characterized to identify mechanisms underlying radioresistance in rectal cancer. Transcriptomic and functional analysis demonstrated significant alterations in multiple molecular pathways, including the cell cycle, DNA repair efficiency and upregulation of oxidative phosphorylation-related genes in radioresistant SW837 rectal cancer cells. Real-time metabolic profiling demonstrated decreased reliance on glycolysis and enhanced mitochondrial spare respiratory capacity in radioresistant SW837 cells when compared to radiosensitive HCT116 cells. Metabolomic profiling of pre-treatment serum samples from rectal cancer patients (n = 52) identified 16 metabolites significantly associated with subsequent pathological response to neoadjuvant chemoradiation therapy. Thirteen of these metabolites were also significantly associated with overall survival. This study demonstrates, for the first time, a role for metabolic reprograming in the radioresistance of rectal cancer in vitro and highlights a potential role for altered metabolites as novel circulating predictive markers of treatment response in rectal cancer patients.
Keywords: glycolysis; metabolism; metabolites; oxidative phosphorylation; radioresistance; rectal cancer.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- National Cancer Registry Ireland (NCRI) Cancer Incidence Projections for Ireland 2020–2045. National Cancer Registry; Cork, Ireland: 2019.
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

