Can Electronegative LDL Act as a Multienzymatic Complex?
- PMID: 37108253
- PMCID: PMC10138509
- DOI: 10.3390/ijms24087074
Can Electronegative LDL Act as a Multienzymatic Complex?
Abstract
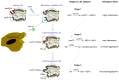
Electronegative LDL (LDL(-)) is a minor form of LDL present in blood for which proportions are increased in pathologies with increased cardiovascular risk. In vitro studies have shown that LDL(-) presents pro-atherogenic properties, including a high susceptibility to aggregation, the ability to induce inflammation and apoptosis, and increased binding to arterial proteoglycans; however, it also shows some anti-atherogenic properties, which suggest a role in controlling the atherosclerotic process. One of the distinctive features of LDL(-) is that it has enzymatic activities with the ability to degrade different lipids. For example, LDL(-) transports platelet-activating factor acetylhydrolase (PAF-AH), which degrades oxidized phospholipids. In addition, two other enzymatic activities are exhibited by LDL(-). The first is type C phospholipase activity, which degrades both lysophosphatidylcholine (LysoPLC-like activity) and sphingomyelin (SMase-like activity). The second is ceramidase activity (CDase-like). Based on the complementarity of the products and substrates of these different activities, this review speculates on the possibility that LDL(-) may act as a sort of multienzymatic complex in which these enzymatic activities exert a concerted action. We hypothesize that LysoPLC/SMase and CDase activities could be generated by conformational changes in apoB-100 and that both activities occur in proximity to PAF-AH, making it feasible to discern a coordinated action among them.
Keywords: LDL aggregation; ceramidase; electronegative LDL; inflammation; low-density lipoprotein; modified LDL; phospholipase C; platelet-activating factor acetylhydrolase; sphingomyelinase.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
References
-
- Kruth H.S., Jones N.L., Huang W., Zhao B., Ishii I., Chang J., Combs C.A., Malide D., Zhang W.-Y. Macropinocytosis Is the Endocytic Pathway That Mediates Macrophage Foam Cell Formation with Native Low Density Lipoprotein. J. Biol. Chem. 2005;280:2352–2360. doi: 10.1074/jbc.M407167200. - DOI - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

