Deficiency of AP1 Complex Ap1g1 in Zebrafish Model Led to Perturbation of Neurodevelopment, Female and Male Fertility; New Insight to Understand Adaptinopathies
- PMID: 37108275
- PMCID: PMC10138411
- DOI: 10.3390/ijms24087108
Deficiency of AP1 Complex Ap1g1 in Zebrafish Model Led to Perturbation of Neurodevelopment, Female and Male Fertility; New Insight to Understand Adaptinopathies
Abstract
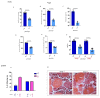
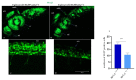
In vertebrates, two homologous heterotetrameric AP1 complexes regulate the intracellular protein sorting via vesicles. AP-1 complexes are ubiquitously expressed and are composed of four different subunits: γ, β1, μ1 and σ1. Two different complexes are present in eukaryotic cells, AP1G1 (contains γ1 subunit) and AP1G2 (contains γ2 subunit); both are indispensable for development. One additional tissue-specific isoform exists for μ1A, the polarized epithelial cells specific to μ1B; two additional tissue-specific isoforms exist for σ1A: σ1B and σ1C. Both AP1 complexes fulfil specific functions at the trans-Golgi network and endosomes. The use of different animal models demonstrated their crucial role in the development of multicellular organisms and the specification of neuronal and epithelial cells. Ap1g1 (γ1) knockout mice cease development at the blastocyst stage, while Ap1m1 (μ1A) knockouts cease during mid-organogenesis. A growing number of human diseases have been associated with mutations in genes encoding for the subunits of adaptor protein complexes. Recently, a new class of neurocutaneous and neurometabolic disorders affecting intracellular vesicular traffic have been referred to as adaptinopathies. To better understand the functional role of AP1G1 in adaptinopathies, we generated a zebrafish ap1g1 knockout using CRISPR/Cas9 genome editing. Zebrafish ap1g1 knockout embryos cease their development at the blastula stage. Interestingly, heterozygous females and males have reduced fertility and showed morphological alterations in the brain, gonads and intestinal epithelium. An analysis of mRNA profiles of different marker proteins and altered tissue morphologies revealed dysregulated cadherin-mediated cell adhesion. These data demonstrate that the zebrafish model organism enables us to study the molecular details of adaptinopathies and thus also develop treatment strategies.
Keywords: CRISPR/Cas9 technique; adaptinopathies; embryonic development; intracellular vesicular trafficking.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Analysis of three μ1-AP1 subunits during zebrafish development.Dev Dyn. 2014 Feb;243(2):299-314. doi: 10.1002/dvdy.24071. Epub 2013 Dec 2. Dev Dyn. 2014. PMID: 24123392
-
γ2 and γ1AP-1 complexes: Different essential functions and regulatory mechanisms in clathrin-dependent protein sorting.Eur J Cell Biol. 2017 Jun;96(4):356-368. doi: 10.1016/j.ejcb.2017.03.008. Epub 2017 Mar 27. Eur J Cell Biol. 2017. PMID: 28372831
-
Characterization of the AP-1 μ1A and μ1B adaptins in zebrafish (Danio rerio).Dev Dyn. 2010 Sep;239(9):2404-12. doi: 10.1002/dvdy.22372. Dev Dyn. 2010. PMID: 20652956
-
Physiological roles of clathrin adaptor AP complexes: lessons from mutant animals.J Biochem. 2006 Jun;139(6):943-8. doi: 10.1093/jb/mvj120. J Biochem. 2006. PMID: 16788044 Review.
-
The role of AP-4 in cargo export from the trans-Golgi network and hereditary spastic paraplegia.Biochem Soc Trans. 2020 Oct 30;48(5):1877-1888. doi: 10.1042/BST20190664. Biochem Soc Trans. 2020. PMID: 33084855 Review.
Cited by
-
OTUD1 regulates cytokine expression and related pathways in goose fatty liver by promoting deubiquitination of its target proteins.Poult Sci. 2024 Dec;103(12):104382. doi: 10.1016/j.psj.2024.104382. Epub 2024 Oct 4. Poult Sci. 2024. PMID: 39437555 Free PMC article.
-
Identification of Novel Genetic Loci Involved in Testis Traits of the Jiangxi Local Breed Based on GWAS Analyses.Genes (Basel). 2025 May 27;16(6):637. doi: 10.3390/genes16060637. Genes (Basel). 2025. PMID: 40565529 Free PMC article.
References
-
- Zizioli D., Geumann C., Kratzke M., Mishra R., Borsani G., Finazzi D., Candiello E., Schu P. γ2AP-1 and γ1AP-1 complexes: Different essential functions and regulatory mechanisms in clathrin-dependent protein sorting. Eur. J. Cell Biol. 2017;96:356–368. doi: 10.1016/j.ejcb.2017.03.008. - DOI - PubMed
-
- Hase K., Nakatsu F., Ohmae M., Sugihara K., Shioda N., Takahashi D., Obata Y., Furusawa Y., Fujimura Y., Yamashita T., et al. AP1B-mediated protein sorting regulates polarity and proliferation of intestinal epithelial cells in mice. Gastroenterology. 2013;145:625–635. doi: 10.1053/j.gastro.2013.05.013. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

