Alveolar-Capillary Barrier Protection In Vitro: Lung Cell Type-Specific Effects and Molecular Mechanisms Induced by 1α, 25-Dihydroxyvitamin D3
- PMID: 37108455
- PMCID: PMC10138495
- DOI: 10.3390/ijms24087298
Alveolar-Capillary Barrier Protection In Vitro: Lung Cell Type-Specific Effects and Molecular Mechanisms Induced by 1α, 25-Dihydroxyvitamin D3
Abstract
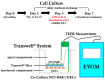
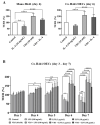
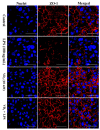
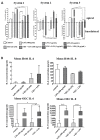
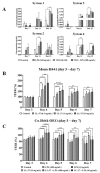
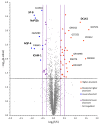
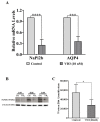
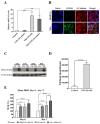
Low serum levels of 1α, 25-dihydroxyvitamin D3 (VD3) are associated with a higher mortality in trauma patients with sepsis or ARDS. However, the molecular mechanisms behind this observation are not yet understood. VD3 is known to stimulate lung maturity, alveolar type II cell differentiation, or pulmonary surfactant synthesis and guides epithelial defense during infection. In this study, we investigated the impact of VD3 on the alveolar-capillary barrier in a co-culture model of alveolar epithelial cells and microvascular endothelial cells respectively in the individual cell types. After stimulation with bacterial LPS (lipopolysaccharide), gene expression of inflammatory cytokines, surfactant proteins, transport proteins, antimicrobial peptide, and doublecortin-like kinase 1 (DCLK1) were analyzed by real-time PCR, while corresponding proteins were evaluated by ELISA, immune-fluorescence, or Western blot. The effect of VD3 on the intracellular protein composition in H441 cells was analyzed by quantitative liquid chromatography-mass spectrometry-based proteomics. VD3 effectively protected the alveolar-capillary barrier against LPS treatment, as indicated by TEER measurement and morphological assessment. VD3 did not inhibit the IL-6 secretion by H441 and OEC but restricted the diffusion of IL-6 to the epithelial compartment. Further, VD3 could significantly suppress the surfactant protein A expression induced in the co-culture system by LPS treatment. VD3 induced high levels of the antimicrobial peptide LL-37, which counteracted effects by LPS and strengthened the barrier. Quantitative proteomics identified VD3-dependent protein abundance changes ranging from constitutional extracellular matrix components and surfactant-associated proteins to immune-regulatory molecules. DCLK1, as a newly described target molecule for VD3, was prominently stimulated by VD3 (10 nM) and seems to influence the alveolar-epithelial cell barrier and regeneration.
Keywords: DCLK1; LC-MS; LPS; VD3; alveolar epithelial cells; co-culture; lung barrier; quantitative proteomics.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Site-specific and endothelial-mediated dysfunction of the alveolar-capillary barrier in response to lipopolysaccharides.J Cell Mol Med. 2018 Feb;22(2):982-998. doi: 10.1111/jcmm.13421. Epub 2017 Dec 5. J Cell Mol Med. 2018. PMID: 29210175 Free PMC article.
-
Role of DCLK1/Hippo pathway in type II alveolar epithelial cells differentiation in acute respiratory distress syndrome.Mol Med. 2023 Nov 23;29(1):159. doi: 10.1186/s10020-023-00760-0. Mol Med. 2023. PMID: 37996782 Free PMC article.
-
Pulmonary administration of 1,25-dihydroxyvitamin D3 to the lungs induces alveolar regeneration in a mouse model of chronic obstructive pulmonary disease.J Control Release. 2016 Jul 10;233:191-7. doi: 10.1016/j.jconrel.2016.05.006. Epub 2016 May 7. J Control Release. 2016. PMID: 27164543
-
Anti-inflammatory effects of 1,25-dihydroxyvitamin D3 in monocytes cultured in serum from patients with type 2 diabetes mellitus and diabetic nephropathy with uremia via Toll-like receptor 4 and nuclear factor-κB p65.Mol Med Rep. 2015 Dec;12(6):8215-22. doi: 10.3892/mmr.2015.4482. Epub 2015 Oct 23. Mol Med Rep. 2015. PMID: 26497229
-
Alveolar-Capillary Membrane-Related Pulmonary Cells as a Target in Endotoxin-Induced Acute Lung Injury.Int J Mol Sci. 2019 Feb 15;20(4):831. doi: 10.3390/ijms20040831. Int J Mol Sci. 2019. PMID: 30769918 Free PMC article. Review.
Cited by
-
Proteomic profiling of over 13,000 proteins across ten mouse tissues using multi-dimensional peptide pre-fractionation.J Proteomics. 2025 Aug 15;319:105484. doi: 10.1016/j.jprot.2025.105484. Epub 2025 Jun 18. J Proteomics. 2025. PMID: 40541929
-
Vitamin D: A Nutraceutical Supplement at the Crossroad Between Respiratory Infections and COVID-19.Int J Mol Sci. 2025 Mar 12;26(6):2550. doi: 10.3390/ijms26062550. Int J Mol Sci. 2025. PMID: 40141190 Free PMC article. Review.
-
A ventilated perfused lung model platform to dissect the response of the lungs to viral infection.Trends Biotechnol. 2025 Jul;43(7):1714-1742. doi: 10.1016/j.tibtech.2025.03.012. Epub 2025 Apr 24. Trends Biotechnol. 2025. PMID: 40280814
References
-
- Takahashi S., Maeda T., Sano Y., Nishihara H., Takeshita Y., Shimizu F., Kanda T. Active form of vitamin D directly protects the blood-brain barrier in multiple sclerosis. Clin. Exp. Neuroimmunol. 2017;8:244–254. doi: 10.1111/cen3.12398. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

