Transcriptomic Analysis Reveals Key Roles of (p)ppGpp and DksA in Regulating Metabolism and Chemotaxis in Yersinia enterocolitica
- PMID: 37108773
- PMCID: PMC10142893
- DOI: 10.3390/ijms24087612
Transcriptomic Analysis Reveals Key Roles of (p)ppGpp and DksA in Regulating Metabolism and Chemotaxis in Yersinia enterocolitica
Abstract
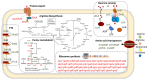
The stringent response is a rapid response system that is ubiquitous in bacteria, allowing them to sense changes in the external environment and undergo extensive physiological transformations. However, the regulators (p)ppGpp and DksA have extensive and complex regulatory patterns. Our previous studies demonstrated that (p)ppGpp and DksA in Yersinia enterocolitica positively co-regulated motility, antibiotic resistance, and environmental tolerance but had opposite roles in biofilm formation. To reveal the cellular functions regulated by (p)ppGpp and DksA comprehensively, the gene expression profiles of wild-type, ΔrelA, ΔrelAΔspoT, and ΔdksAΔrelAΔspoT strains were compared using RNA-Seq. Results showed that (p)ppGpp and DksA repressed the expression of ribosomal synthesis genes and enhanced the expression of genes involved in intracellular energy and material metabolism, amino acid transport and synthesis, flagella formation, and the phosphate transfer system. Additionally, (p)ppGpp and DksA inhibited amino acid utilization (such as arginine and cystine) and chemotaxis in Y. enterocolitica. Overall, the results of this study unraveled the link between (p)ppGpp and DksA in the metabolic networks, amino acid utilization, and chemotaxis in Y. enterocolitica and enhanced the understanding of stringent responses in Enterobacteriaceae.
Keywords: (p)ppGpp; DksA; Yersinia enterocolitica; chemotaxis; stringent response.
Conflict of interest statement
The authors declare no conflict of interest.
Figures





Similar articles
-
Stringent Response Factor DksA Contributes to Fatty Acid Degradation Function to Influence Cell Membrane Stability and Polymyxin B Resistance of Yersinia enterocolitica.Int J Mol Sci. 2023 Jul 26;24(15):11951. doi: 10.3390/ijms241511951. Int J Mol Sci. 2023. PMID: 37569327 Free PMC article.
-
Similar and Divergent Roles of Stringent Regulator (p)ppGpp and DksA on Pleiotropic Phenotype of Yersinia enterocolitica.Microbiol Spectr. 2022 Dec 21;10(6):e0205522. doi: 10.1128/spectrum.02055-22. Epub 2022 Nov 21. Microbiol Spectr. 2022. PMID: 36409141 Free PMC article.
-
Stringent response regulators (p)ppGpp and DksA positively regulate virulence and host adaptation of Xanthomonas citri.Mol Plant Pathol. 2019 Nov;20(11):1550-1565. doi: 10.1111/mpp.12865. Epub 2019 Oct 17. Mol Plant Pathol. 2019. PMID: 31621195 Free PMC article.
-
Direct binding targets of the stringent response alarmone (p)ppGpp.Mol Microbiol. 2012 Sep;85(6):1029-43. doi: 10.1111/j.1365-2958.2012.08177.x. Epub 2012 Aug 2. Mol Microbiol. 2012. PMID: 22812515 Review.
-
Transcriptional Responses to ppGpp and DksA.Annu Rev Microbiol. 2018 Sep 8;72:163-184. doi: 10.1146/annurev-micro-090817-062444. Annu Rev Microbiol. 2018. PMID: 30200857 Free PMC article. Review.
Cited by
-
(p)ppGpp and DksA play a crucial role in reducing the efficacy of β-lactam antibiotics by modulating bacterial membrane permeability.Microbiol Spectr. 2025 Apr;13(4):e0116924. doi: 10.1128/spectrum.01169-24. Epub 2025 Feb 24. Microbiol Spectr. 2025. PMID: 39992161 Free PMC article.
-
In-host evolution of Yersinia enterocolitica during a chronic human infection.Nat Commun. 2025 Jul 1;16(1):5637. doi: 10.1038/s41467-025-60782-6. Nat Commun. 2025. PMID: 40595576 Free PMC article.
-
Stringent Response Factor DksA Contributes to Fatty Acid Degradation Function to Influence Cell Membrane Stability and Polymyxin B Resistance of Yersinia enterocolitica.Int J Mol Sci. 2023 Jul 26;24(15):11951. doi: 10.3390/ijms241511951. Int J Mol Sci. 2023. PMID: 37569327 Free PMC article.
-
The stringent response regulates the poly-β-hydroxybutyrate (PHB) synthesis in Azotobacter vinelandii.PLoS One. 2024 Apr 4;19(4):e0299640. doi: 10.1371/journal.pone.0299640. eCollection 2024. PLoS One. 2024. PMID: 38574051 Free PMC article.
-
Metagenomic and metabolomic analysis of changes in intestinal contents of rainbow trout (Oncorhynchus mykiss) infected with infectious hematopoietic necrosis virus at different culture water temperatures.Front Microbiol. 2023 Oct 16;14:1275649. doi: 10.3389/fmicb.2023.1275649. eCollection 2023. Front Microbiol. 2023. PMID: 37908544 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

