Can Neutrophils Prevent Nosocomial Pneumonia after Serious Injury?
- PMID: 37108790
- PMCID: PMC10141656
- DOI: 10.3390/ijms24087627
Can Neutrophils Prevent Nosocomial Pneumonia after Serious Injury?
Abstract
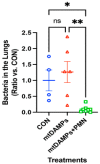
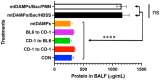
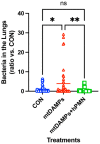
Nosocomial pneumonia is a leading cause of critical illness and mortality among seriously injured trauma patients. However, the link between injury and the development of nosocomial pneumonia is still not well recognized. Our work strongly suggests that mitochondrial damage-associated molecular patterns (mtDAMPs), especially mitochondrial formyl peptides (mtFPs) released by tissue injury, play a significant role in developing nosocomial pneumonia after a serious injury. Polymorphonuclear leukocytes (neutrophils, PMN) migrate toward the injury site by detecting mtFPs through formyl peptide receptor 1 (FPR1) to fight/contain bacterial infection and clean up debris. Activation of FPR1 by mtFPs enables PMN to reach the injury site; however, at the same time it leads to homo- and heterologous desensitization/internalization of chemokine receptors. Thus, PMN are not responsive to secondary infections, including those from bacteria-infected lungs. This may enable a progression of bacterial growth in the lungs and nosocomial pneumonia. We propose that the intratracheal application of exogenously isolated PMN may prevent pneumonia coupled with a serious injury.
Keywords: FPR1; infection; injury; innate immunity; neutrophils; nosocomial pneumonia; trauma.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
A subset of five human mitochondrial formyl peptides mimics bacterial peptides and functionally deactivates human neutrophils.J Trauma Acute Care Surg. 2018 Nov;85(5):936-943. doi: 10.1097/TA.0000000000001971. J Trauma Acute Care Surg. 2018. PMID: 29787548
-
Circulating mitochondrial N-formyl peptides contribute to secondary nosocomial infection in patients with septic shock.Proc Natl Acad Sci U S A. 2021 Apr 27;118(17):e2018538118. doi: 10.1073/pnas.2018538118. Proc Natl Acad Sci U S A. 2021. PMID: 33888581 Free PMC article.
-
Direct Airway Instillation of Neutrophils Overcomes Chemotactic Deficits Induced by Injury.Shock. 2021 Jul 1;56(1):119-124. doi: 10.1097/SHK.0000000000001691. Shock. 2021. PMID: 33181622 Free PMC article.
-
The potential impacts of formyl peptide receptor 1 in inflammatory diseases.Front Biosci (Elite Ed). 2016 Jun 1;8(3):436-49. doi: 10.2741/E778. Front Biosci (Elite Ed). 2016. PMID: 27100350 Review.
-
Role of Mitochondria-Derived Danger Signals Released After Injury in Systemic Inflammation and Sepsis.Antioxid Redox Signal. 2021 Nov 20;35(15):1273-1290. doi: 10.1089/ars.2021.0052. Epub 2021 May 25. Antioxid Redox Signal. 2021. PMID: 33847158 Free PMC article. Review.
Cited by
-
Construction of a prediction model for pneumonia in children undergoing neurosurgery based on regular clinical laboratory tests and baseline information.Front Pediatr. 2025 Aug 6;13:1638012. doi: 10.3389/fped.2025.1638012. eCollection 2025. Front Pediatr. 2025. PMID: 40843073 Free PMC article.
-
The immune suppressive properties of damage associated molecular patterns in the setting of sterile traumatic injury.Front Immunol. 2023 Aug 15;14:1239683. doi: 10.3389/fimmu.2023.1239683. eCollection 2023. Front Immunol. 2023. PMID: 37662933 Free PMC article. Review.
-
C/EBPε and its acetylation in PMN enhance the tolerance to trauma.Clin Exp Immunol. 2025 Jan 21;219(1):uxae061. doi: 10.1093/cei/uxae061. Clin Exp Immunol. 2025. PMID: 39028614 Free PMC article.
-
Transcriptomics reveals the underlying mechanism of Prostaglandin E1 in improving severe pneumonia.BMC Pulm Med. 2025 Aug 14;25(1):389. doi: 10.1186/s12890-025-03847-y. BMC Pulm Med. 2025. PMID: 40804382 Free PMC article.
References
-
- Chevret S., Hemmer M., Carlet J., Langer M. Incidence and risk factors of pneumonia acquired in intensive care units. Results from a multicenter prospective study on 996 patients. European Cooperative Group on Nosocomial Pneumonia. Intensive Care Med. 1993;19:256–264. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

