Role of Oritavancin in the Treatment of Infective Endocarditis, Catheter- or Device-Related Infections, Bloodstream Infections, and Bone and Prosthetic Joint Infections in Humans: Narrative Review and Possible Developments
- PMID: 37109488
- PMCID: PMC10143027
- DOI: 10.3390/life13040959
Role of Oritavancin in the Treatment of Infective Endocarditis, Catheter- or Device-Related Infections, Bloodstream Infections, and Bone and Prosthetic Joint Infections in Humans: Narrative Review and Possible Developments
Abstract
Oritavancin is a long-acting lipoglycopeptide with in vitro activity against Gram-positive pathogens, as well as good bactericidal activity and sterilisation ability in biofilm. It has been approved for acute bacterial skin and skin structure infections (ABSSSI), but recent reports have demonstrated possible off-label uses, such as for vancomycin resistant enterococci (VRE), deep-seated infections including those involving prosthetic material and invasive infections. The aim of this work is to review the uses of oritavancin outside of ABSSSI, focusing on its real-life applications on infective endocarditis, catheter- or device-related infections, bloodstream infections, and bone and prosthetic joint infections in humans, as well as possible future applications. We performed a narrative review, collecting the literature published between 1 December 2002 and 1 November 2022 on PubMed and the Cochrane Library using the term 'oritavancin'. Available studies have shown how effective it is in different settings, suggesting an opportunity for step-down strategies or outpatient management of infections requiring a long duration of antibiotic treatment. So far, evidence is still scarce, and limited to a few studies and case reports, mostly focusing on Staphylococcus aureus as the major isolate. Concerns about fluid intake for dilution and interaction with coagulation markers also need to be taken into account. Further studies are required in order to assess the safety and effectiveness of Oritavancin in vascular, prosthetic, or device-related infections, as well as in resistant Gram-positive bacteria or enterococcal infections.
Keywords: Gram-positive; device infections; enterococcal infections; long-acting; oritavancin; prosthetic infections; review; vascular infections.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
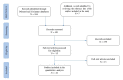

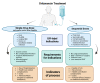
References
-
- Sintesi di Tenkasi e Perché è Autorizzato Nell’unione Europea (UE) [(accessed on 5 November 2022)]. Available online: https://www.ema.europa.eu/en/documents/overview/tenkasi-previously-orbac....
-
- Highlights of Prescribing Information. [(accessed on 5 November 2022)]; Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/206334s005lbl.pdf.
Publication types
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

