Effects of Topical or Intravitreal Application of Anti-Vascular Endothelial Growth Factor on Density of Intestinal Blood Vessels of Mice
- PMID: 37109767
- PMCID: PMC10143914
- DOI: 10.3390/medicina59040809
Effects of Topical or Intravitreal Application of Anti-Vascular Endothelial Growth Factor on Density of Intestinal Blood Vessels of Mice
Abstract
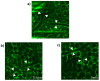
Background and Objectives: Anti-vascular endothelial growth factor (anti-VEGF) therapy has become the first-line treatment for diabetic macular edema. However, it is still not clear whether anti-VEGF agents act on systemic blood vessels. The aim of this study is to determine whether a direct topical application or intravitreal injection of anti-VEGF will change the intestinal blood vessels of mice. Materials and Methods: C57BL/6 mice were laparotomied under deep anesthesia, and the blood vessels on the surface of the intestines were exposed, examined, and photographed through a dissecting microscope. Vascular changes were evaluated before and at 1, 5, and 15 min after the topical application of 50 µL of the different anti-VEGF agents onto the surface of the intestine (group S) or after the intravitreal injection (group V). The vascular density (VD) was determined for five mice in each group before and after 40 μg/μL of aflibercept (Af), or 25 μg/μL of bevacizumab (Be), or 10 μg/μL of ranibizumab (Ra) were applied. Endothelin-1 (ET1), a potent vasoconstrictor, was used as a positive control, and phosphate-buffered saline (PBS) was used as a control. Results: For group S, no significant changes were observed after PBS (baseline, 1, 5, and 15 min: 46.3, 44.5, 44.8, and 43.2%), Be (46.1, 46.7, 46.7, and 46.3%), Ra (44.7, 45.0, 44.7, and 45.6%), and Af (46.5, 46.2, 45.9, and 46.1%, repeated ANOVA) were applied topically. Significant decreases in the VD were observed after ET1 (46.7, 28.1, 32.1, and 34.0%, p < 0.05) was topically applied. For group V, no significant differences were observed for all anti-VEGF agents. Conclusions: The topical application or intravitreal injections of anti-VEGF agents do not cause a change in the VD of the intestinal vessels, which may be related to its safety.
Keywords: anti-vascular endothelial growth factor agents; direct titration; intravitreal injection; vasoconstriction.
Conflict of interest statement
Atsushi Ichio, Yuhki Koike and Yuji Toiyama have: None. Masahiko Sugimoto has: [F] Alcon Pharma (class III), Bayer (class III), Chugai (class III); [R] Kowa Pharma (class II), Senjyu Pharma (class II), Daiichi Yakuhin Sangyo (class II), Bayer (class II), Santen (class II), Wakamoto Pharma (class II). Mineo Kondo has: [F] Alcon Pharma (class III), Bayer (class III), AMO (class III), Ohtsuka (class III), Kowa (class III), Santen (class III), Senjyu Pharma (class III), Pfizer (class III), HOYA (class III), Novartis Pharma (class III); [R] Santen (class III), Sanwa Kagaku (class III), Novartis Pharma (class III), Alcon Pharma (class III), Bayer (class II), Avi (class II), AMO (class II), Ohtsuka (class II), Kowa (class III), Sanofi (class II), Senjyu Pharma (class II), Tanabe-Mitsui Pharma (class II), Pfizer (class II), Rhoto (class II); [C] Senjyu (class III), Ono (class III), Rhort (class III).
Figures



References
-
- Nguyen Q.D., Brown D.M., Marcus D.M., Boyer D.S., Patel S., Feiner L., Gibson A., Sy J., Rundle A.C., Hopkins J.J., et al. Ranibizumab for diabetic macular edema: Results from 2 phase III randomized trials: RISE and RIDE. Ophthalmology. 2012;119:789–801. doi: 10.1016/j.ophtha.2011.12.039. - DOI - PubMed
-
- Brown D.M., Schmidt-Erfurth U., Do D.V., Holz F.G., Boyer D.S., Midena E., Heier J.S., Terasaki H., Kaiser P.K., Marcus D.M., et al. Intravitreal aflibercept for diabetic macular edema: 100-week results from the VISTA and VIVID studies. Ophthalmology. 2015;122:2044–2052. doi: 10.1016/j.ophtha.2015.06.017. - DOI - PubMed
-
- Kodjikian L., Bellocq D., Bandello F., Loewenstein A., Chakravarthy U., Koh A., Augustin A., de Smet M.D., Chhablani J., Tufail A., et al. First-line treatment algorithm and guidelines in center-involving diabetic macular edema. Eur. J. Ophthalmol. 2019;29:573–584. doi: 10.1177/1120672119857511. - DOI - PubMed
-
- Zarbin M.A. Anti-VEGF Agents and the Risk of Arteriothrombotic Events. Asia-Pac. J. Ophthalmol. 2018;7:63–67. - PubMed
-
- Azzolini C., Pagani I.S., Pirrone C., Borroni D., Donati S., Al Oum M., Pigni D., Chiaravalli A.M., Vinciguerra R., Simonelli F., et al. Expression of VEGF-A, Otx homeobox and p53 family genes in proliferative vitreoretinopathy. Mediat. Inflamm. 2013;2013:857380. doi: 10.1155/2013/857380. - DOI - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

