Electrochemical and Photoelectrochemical Immunosensors for the Detection of Ovarian Cancer Biomarkers
- PMID: 37112447
- PMCID: PMC10142013
- DOI: 10.3390/s23084106
Electrochemical and Photoelectrochemical Immunosensors for the Detection of Ovarian Cancer Biomarkers
Abstract
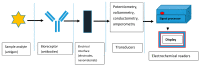
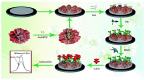
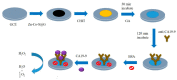
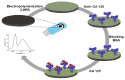
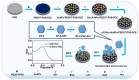
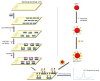
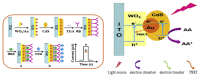
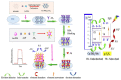
Photoelectrochemical (PEC) sensing is an emerging technological innovation for monitoring small substances/molecules in biological or non-biological systems. In particular, there has been a surge of interest in developing PEC devices for determining molecules of clinical significance. This is especially the case for molecules that are markers for serious and deadly medical conditions. The increased interest in PEC sensors to monitor such biomarkers can be attributed to the many apparent advantages of the PEC system, including an enhanced measurable signal, high potential for miniaturization, rapid testing, and low cost, amongst others. The growing number of published research reports on the subject calls for a comprehensive review of the various findings. This article is a review of studies on electrochemical (EC) and PEC sensors for ovarian cancer biomarkers in the last seven years (2016-2022). EC sensors were included because PEC is an improved EC; and a comparison of both systems has, expectedly, been carried out in many studies. Specific attention was given to the different markers of ovarian cancer and the EC/PEC sensing platforms developed for their detection/quantification. Relevant articles were sourced from the following databases: Scopus, PubMed Central, Web of Science, Science Direct, Academic Search Complete, EBSCO, CORE, Directory of open Access Journals (DOAJ), Public Library of Science (PLOS), BioMed Central (BMC), Semantic Scholar, Research Gate, SciELO, Wiley Online Library, Elsevier and SpringerLink.
Keywords: cancer biomarkers; electrochemical sensing; ovarian cancer; photoelectrochemical immunosensors.
Conflict of interest statement
The authors declare that there is no conflict of interest.
Figures













Similar articles
-
Ultrasensitive photoelectrochemical microcystin-LR immunosensor using carboxyl-functionalized graphene oxide enhanced gold nanoclusters for signal amplification.Anal Chim Acta. 2021 Nov 15;1185:339078. doi: 10.1016/j.aca.2021.339078. Epub 2021 Sep 18. Anal Chim Acta. 2021. PMID: 34711309
-
Signal amplification strategies in photoelectrochemical sensing of carcinoembryonic antigen.Biosens Bioelectron. 2024 Oct 15;262:116543. doi: 10.1016/j.bios.2024.116543. Epub 2024 Jun 28. Biosens Bioelectron. 2024. PMID: 38963951 Review.
-
PB@PDA nanocomposites as nanolabels and signal reporters for separate-type cathodic photoelectrochemical immunosensors in the detection of carcinoembryonic antigens.Talanta. 2023 Mar 1;254:124134. doi: 10.1016/j.talanta.2022.124134. Epub 2022 Nov 23. Talanta. 2023. PMID: 36450179
-
Immunosensors Based on Nanomaterials for Detection of Tumor Markers.J Biomed Nanotechnol. 2018 Jan 1;14(1):44-65. doi: 10.1166/jbn.2018.2505. J Biomed Nanotechnol. 2018. PMID: 29463365 Review.
-
Recent advances in microfluidic-based photoelectrochemical (PEC) sensing platforms for biomedical applications.Mikrochim Acta. 2025 Apr 14;192(5):297. doi: 10.1007/s00604-025-07135-8. Mikrochim Acta. 2025. PMID: 40229472 Review.
Cited by
-
Recent advances in aptamer-based biosensing technology for isolation and detection of extracellular vesicles.Front Cell Dev Biol. 2025 Jul 23;13:1555687. doi: 10.3389/fcell.2025.1555687. eCollection 2025. Front Cell Dev Biol. 2025. PMID: 40772226 Free PMC article. Review.
-
Ceramic Perovskite-Based Photoelectrochemical Platform for 2‑(3,4-Dihydroxyphenyl)ethylamine Detection with Enhanced Sensitivity.ACS Omega. 2025 Jul 4;10(27):29256-29266. doi: 10.1021/acsomega.5c02185. eCollection 2025 Jul 15. ACS Omega. 2025. PMID: 40687042 Free PMC article.
-
Modern Emerging Biosensing Methodologies for the Early Diagnosis and Screening of Ovarian Cancer.Biosensors (Basel). 2025 Mar 21;15(4):203. doi: 10.3390/bios15040203. Biosensors (Basel). 2025. PMID: 40277517 Free PMC article. Review.
-
A novel electrochemical immunosensor based on biomaterials for detecting carcinoembryonic antigen biomarker in serum samples.Sci Rep. 2025 Jul 14;15(1):25396. doi: 10.1038/s41598-025-09547-1. Sci Rep. 2025. PMID: 40659702 Free PMC article.
References
-
- Arjmand M., Avval F. Clinical biomarkers for detection of ovarian cancer. J. Mol. Cancer. 2019;2:3–7.
-
- Centers for Disease Control and Prevention Basic Information About Ovarian Cancer. [(accessed on 26 June 2022)];2021 Available online: www.cdc.gov/cancer/ovarian/basic_info/index.htm.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

