Impact of Borna Disease Virus Infection on the Transcriptome of Differentiated Neuronal Cells and Its Modulation by Antiviral Treatment
- PMID: 37112922
- PMCID: PMC10145824
- DOI: 10.3390/v15040942
Impact of Borna Disease Virus Infection on the Transcriptome of Differentiated Neuronal Cells and Its Modulation by Antiviral Treatment
Abstract
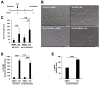
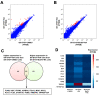
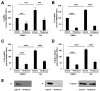
Borna disease virus (BoDV-1) is a highly neurotropic RNA virus that causes neurobehavioral disturbances such as abnormal social activities and memory impairment. Although impairments in the neural circuits caused by BoDV-1 infection induce these disturbances, the molecular basis remains unclear. Furthermore, it is unknown whether anti-BoDV-1 treatments can attenuate BoDV-1-mediated transcriptomic changes in neuronal cells. In this study, we investigated the effects of BoDV-1 infection on neuronal differentiation and the transcriptome of differentiated neuronal cells using persistently BoDV-1-infected cells. Although BoDV-1 infection did not have a detectable effect on intracellular neuronal differentiation processes, differentiated neuronal cells exhibited transcriptomic changes in differentiation-related genes. Some of these transcriptomic changes, such as the decrease in the expression of apoptosis-related genes, were recovered by anti-BoDV-1 treatment, while alterations in the expression of other genes remained after treatment. We further demonstrated that a decrease in cell viability induced by differentiation processes in BoDV-1-infected cells can be relieved with anti-BoDV-1 treatment. This study provides fundamental information regarding transcriptomic changes after BoDV-1 infection and the treatment in neuronal cells.
Keywords: Borna disease virus; antiviral; differentiation; gene expression; neuronal cells.
Conflict of interest statement
The authors declare no conflict of interest.
Figures


Similar articles
-
Suppression of Borna Disease Virus Replication during Its Persistent Infection Using the CRISPR/Cas13b System.Int J Mol Sci. 2024 Mar 20;25(6):3523. doi: 10.3390/ijms25063523. Int J Mol Sci. 2024. PMID: 38542493 Free PMC article.
-
The regulation of persistent Borna disease virus infection by RNA silencing factors in human cells.Biochem Biophys Res Commun. 2023 May 28;658:122-127. doi: 10.1016/j.bbrc.2023.03.069. Epub 2023 Mar 31. Biochem Biophys Res Commun. 2023. PMID: 37030066
-
miR-505 inhibits replication of Borna disease virus 1 via inhibition of HMGB1-mediated autophagy.J Gen Virol. 2022 Jan;103(1). doi: 10.1099/jgv.0.001713. J Gen Virol. 2022. PMID: 35060474
-
Reverse Genetics and Artificial Replication Systems of Borna Disease Virus 1.Viruses. 2022 Oct 12;14(10):2236. doi: 10.3390/v14102236. Viruses. 2022. PMID: 36298790 Free PMC article. Review.
-
Reverse genetics approaches of Borna disease virus: applications in development of viral vectors and preventive vaccines.Curr Opin Virol. 2020 Oct;44:42-48. doi: 10.1016/j.coviro.2020.05.011. Epub 2020 Jul 10. Curr Opin Virol. 2020. PMID: 32659515 Review.
Cited by
-
Suppression of Borna Disease Virus Replication during Its Persistent Infection Using the CRISPR/Cas13b System.Int J Mol Sci. 2024 Mar 20;25(6):3523. doi: 10.3390/ijms25063523. Int J Mol Sci. 2024. PMID: 38542493 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

