Diffusion-tensor magnetic resonance imaging captures increased skeletal muscle fibre diameters in Becker muscular dystrophy
- PMID: 37127427
- PMCID: PMC10235880
- DOI: 10.1002/jcsm.13242
Diffusion-tensor magnetic resonance imaging captures increased skeletal muscle fibre diameters in Becker muscular dystrophy
Abstract
Background: Becker muscular dystrophy (BMD) is an X-linked disorder characterized by slow, progressive muscle damage and muscle weakness. Hallmarks include fibre-size variation and replacement of skeletal muscle with fibrous and adipose tissues, after repeated cycles of regeneration. Muscle histology can detect these features, but the required biopsies are invasive, are difficult to repeat and capture only small muscle volumes. Diffusion-tensor magnetic resonance imaging (DT-MRI) is a potential non-invasive alternative that can calculate muscle fibre diameters when applied with the novel random permeable barrier model (RPBM). In this study, we assessed muscle fibre diameters using DT-MRI in BMD patients and healthy controls and compared these with histology.
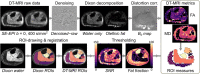
Methods: We included 13 BMD patients and 9 age-matched controls, who underwent water-fat MRI and DT-MRI at multiple diffusion times, allowing RPBM parameter estimation in the lower leg muscles. Tibialis anterior muscle biopsies were taken from the contralateral leg in 6 BMD patients who underwent DT-MRI and from an additional 32 BMD patients and 15 healthy controls. Laminin and Sirius-red stainings were performed to evaluate muscle fibre morphology and fibrosis. Twelve ambulant patients from the MRI cohort underwent the North Star ambulatory assessment, and 6-min walk, rise-from-floor and 10-m run/walk functional tests.
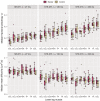
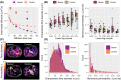
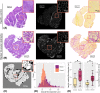
Results: RPBM fibre diameter was significantly larger in BMD patients (P = 0.015): mean (SD) = 68.0 (25.3) μm versus 59.4 (19.2) μm in controls. Inter-muscle differences were also observed (P ≤ 0.002). Both inter- and intra-individual RPBM fibre diameter variability were similar between groups. Laminin staining agreed with the RPBM, showing larger median fibre diameters in patients than in controls: 72.5 (7.9) versus 63.2 (6.9) μm, P = 0.006. However, despite showing similar inter-individual variation, patients showed more intra-individual fibre diameter variability than controls-mean variance (SD) = 34.2 (7.9) versus 21.4 (6.9) μm, P < 0.001-and larger fibrosis areas: median (interquartile range) = 21.7 (5.6)% versus 14.9 (3.4)%, P < 0.001. Despite good overall agreement of RPBM and laminin fibre diameters, they were not associated in patients who underwent DT-MRI and muscle biopsy, perhaps due to lack of colocalization of DT-MRI with biopsy samples.
Conclusions: DT-MRI RPBM metrics agree with histology and can quantify changes in muscle fibre size that are associated with regeneration without the need for biopsies. They therefore show promise as imaging biomarkers for muscular dystrophies.
Keywords: Becker muscular dystrophy; diffusion-tensor MRI; histopathology; immunohistochemistry; skeletal muscle.
© 2023 The Authors. Journal of Cachexia, Sarcopenia and Muscle published by John Wiley & Sons Ltd on behalf of Society on Sarcopenia, Cachexia and Wasting Disorders.
Conflict of interest statement
D.C., T.A.D., L.G.M.H., N.M.V., Z.K., T.T.J.V., M.T.H., S.A., S.G.D., J.J.G.M.V., M.P., V.R. and P.S. report no relevant disclosures. A.A.R. discloses being employed by LUMC, which has patents on exon skipping technology, some of which have been licensed to BioMarin and subsequently sublicensed to Sarepta. As co‐inventor of some of these patents, A.A.R. is entitled to a share of royalties. A.A.R. further discloses being ad hoc consultant for PTC Therapeutics, Sarepta Therapeutics, Regenxbio, Alpha Anomeric, BioMarin Pharmaceuticals Inc., Eisai, Entrada, Takeda, Splicesense, Galapagos and AstraZeneca, with past ad hoc consulting for CRISPR Therapeutics, Summit PLC, Audentes Santhera, Bridge Bio, Global Guidepoint and GLG Consultancy, Grünenthal, Wave and BioClinica. A.A.R. also reports having been a member of the Duchenne Network Steering Committee (BioMarin), being a member of the scientific advisory boards of Eisai, Hybridize Therapeutics, Silence Therapeutics and Sarepta Therapeutics, and being a former member of the scientific advisory boards of ProQR and Philae Pharmaceuticals. LUMC also received speaker honoraria from PTC Therapeutics and BioMarin Pharmaceuticals and funding for contract research from Italfarmaco, Sapreme, Eisai, Galapagos, Synaffix and Alpha Anomeric. A.A.R. received project funding from Sarepta Therapeutics. E.H.N. reports ad hoc consultancies for Wave, Santhera, Regenxbio and PTC, and he worked as investigator of clinical trials of Italfarmaco, NS Pharma, Reveragen, Roche, Wave and Sarepta outside the submitted work. H.E.K. reports research support from Philips Healthcare during the conduct of the study, consultancy for PTC Therapeutics and EspeRare and trial support from ImagingDMD‐UF outside the submitted work. All reimbursements for A.A.R., E.H.N. and H.E.K. were received by the LUMC. No personal financial benefits were received.
Figures

References
-
- Faridian‐Aragh N, Wagner KR, Leung DG, Carrino JA. Magnetic resonance imaging phenotyping of Becker muscular dystrophy. Muscle Nerve 2014;50:962–967. - PubMed
-
- Relaix F, Zammit PS. Satellite cells are essential for skeletal muscle regeneration: the cell on the edge returns centre stage. Development 2012;139:2845–2856. - PubMed
-
- Bradley WG, Jones MZ, Mussini JM, Fawcett PRW. Becker‐type muscular dystrophy. Muscle Nerve 1978;1:111–132. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

