Assessment of Tumor Mutational Burden and Outcomes in Patients With Diverse Advanced Cancers Treated With Immunotherapy
- PMID: 37129893
- PMCID: PMC10155064
- DOI: 10.1001/jamanetworkopen.2023.11181
Assessment of Tumor Mutational Burden and Outcomes in Patients With Diverse Advanced Cancers Treated With Immunotherapy
Abstract
Importance: There are few studies assessing the association of tumor mutational burden (TMB) and clinical outcomes in a large cohort of patients with diverse advanced cancers.
Objective: To clinically validate a TMB biomarker from a next-generation sequencing targeted gene panel assay.
Design, setting, and participants: A prespecified cohort study using the deidentified clinicogenomic Tempus database of patients sequenced between 2018 and 2022, which contained retrospective, observational data originating from 300 cancer sites including 199 community sites and 101 academic sites. Patients with advanced solid tumors across 8 cancer types and more than 20 histologies, sequenced with Tempus xT who were treated with immune checkpoint inhibitors (ICIs) in the first-line or second-line setting were included. Data were analyzed from September 2018 to August 2022.
Exposure: Treatment with US Food and Drug Administration (FDA)-approved antiprogrammed cell death-1/programmed cell death-ligand 1 (PD-1/PD-L1) ICI and/or in combination with a cytotoxic T-lymphocyte-associated protein-4 ICI.
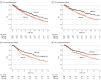
Main outcomes and measures: The primary outcome was the association of tumor mutational burden (TMB) binary category (high [≥10 mut/mb] vs low) with overall survival (OS) in patients treated with ICIs. Secondary outcomes were progression-free survival (PFS), and time to progression (TTP).
Results: In the evaluable cohort of 674 patients, the median (IQR) age was 69.4 (28.6-89.8) years, 271 patients (40.2%) were female, and 435 patients (64.5%) were White. The most common advanced cancers were non-small cell lung cancer (330 patients [49.0%]), followed by bladder cancer (148 patients [22.0%]), and head and neck squamous cell carcinoma (96 patients [14.8%]). Median (IQR) follow-up was 7.2 (3.2-14.1) months. High TMB (TMB-H) cancers (206 patients [30.6%]) were significantly associated with longer OS than low TMB (TMB-L) cancers (hazard ratio [HR], 0.72; upper confidence bound [UCB], 0.91; P = .01). In a prospective subset of 403 patients treated with ICIs after TMB testing, TMB-H cancers (135 patients [33.5%]) were significantly associated with longer OS (HR, 0.61; UCB, 0.84; P = .005), PFS (HR, 0.62; UCB, 0.82; P = .003), and TTP (HR, 0.67; UCB, 0.92; P = .02) than TMB-L cancers. An overall survival benefit was seen regardless of the type of ICI used (pembrolizumab, 339 patients; HR, 0.67; UCB, 0.94; P = .03), other ICIs (64 patients; HR, 0.37; UCB, 0.85; P = .03), and after adjusting for PD-L1 and microsatellite stability status (403 patients; HR = 0.67; UCB, 0.92; P = .02).
Conclusions and relevance: In this cohort study of patients with advanced solid tumors treated with ICIs in diverse clinics, TMB-H cancers were significantly associated with improved clinical outcomes compared with TMB-L cancers.
Conflict of interest statement
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Research Materials
Miscellaneous

