Can we predict who will benefit most from biologics in severe asthma? A post-hoc analysis of two phase 3 trials
- PMID: 37131185
- PMCID: PMC10155396
- DOI: 10.1186/s12931-023-02409-2
Can we predict who will benefit most from biologics in severe asthma? A post-hoc analysis of two phase 3 trials
Abstract
Background: Individualized prediction of treatment response may improve the value proposition of advanced treatment options in severe asthma. This study aimed to investigate the combined capacity of patient characteristics in predicting treatment response to mepolizumab in patients with severe asthma.
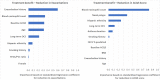
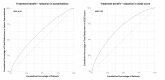
Methods: Patient-level data were pooled from two multinational phase 3 trials of mepolizumab in severe eosinophilic asthma. We fitted penalized regression models to quantify reductions in the rate of severe exacerbations and the 5-item Asthma Control Questionnaire (ACQ5) score. The capacity of 15 covariates towards predicting treatment response was quantified by the Gini index (measuring disparities in treatment benefit) as well as observed treatment benefit within the quintiles of predicted treatment benefit.
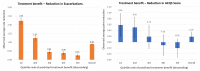
Results: There was marked variability in the ability of patient characteristics to predict treatment response; covariates explained greater heterogeneity in predicting treatment response to asthma control than to exacerbation frequency (Gini index 0.35 v. 0.24). Key predictors for treatment benefit for severe exacerbations included exacerbation history, blood eosinophil count, baseline ACQ5 score and age, and those for symptom control included blood eosinophil count and presence of nasal polyps. Overall, the average reduction in exacerbations was 0.90/year (95%CI, 0.87‒0.92) and average reduction in ACQ5 score was 0.18 (95% CI, 0.02‒0.35). Among the top 20% of patients for predicted treatment benefit, exacerbations were reduced by 2.23/year (95% CI, 2.03‒2.43) and ACQ5 score were reduced by 0.59 (95% CI, 0.19‒0.98). Among the bottom 20% of patients for predicted treatment benefit, exacerbations were reduced by 0.25/year (95% CI, 0.16‒0.34) and ACQ5 by -0.20 (95% CI, -0.51 to 0.11).
Conclusion: A precision medicine approach based on multiple patient characteristics can guide biologic therapy in severe asthma, especially in identifying patients who will not benefit as much from therapy. Patient characteristics had a greater capacity to predict treatment response to asthma control than to exacerbation.
Trial registration: ClinicalTrials.gov number, NCT01691521 (registered September 24, 2012) and NCT01000506 (registered October 23, 2009).
Keywords: Biologics; Mepolizumab; Prediction; Severe asthma; Treatment response.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no conflict of interests.
Figures
Similar articles
-
Severe eosinophilic asthma treated with mepolizumab stratified by baseline eosinophil thresholds: a secondary analysis of the DREAM and MENSA studies.Lancet Respir Med. 2016 Jul;4(7):549-556. doi: 10.1016/S2213-2600(16)30031-5. Epub 2016 May 10. Lancet Respir Med. 2016. PMID: 27177493 Clinical Trial.
-
Evaluation of Potential Continuation Rules for Mepolizumab Treatment of Severe Eosinophilic Asthma.J Allergy Clin Immunol Pract. 2018 May-Jun;6(3):874-882.e4. doi: 10.1016/j.jaip.2017.11.026. Epub 2017 Dec 16. J Allergy Clin Immunol Pract. 2018. PMID: 29258789 Clinical Trial.
-
Baseline blood eosinophil count as a predictor of treatment response to the licensed dose of mepolizumab in severe eosinophilic asthma.Respir Med. 2019 Nov;159:105806. doi: 10.1016/j.rmed.2019.105806. Epub 2019 Nov 3. Respir Med. 2019. PMID: 31751853
-
Impact of baseline clinical asthma characteristics on the response to mepolizumab: a post hoc meta-analysis of two Phase III trials.Respir Res. 2021 Jun 22;22(1):184. doi: 10.1186/s12931-021-01767-z. Respir Res. 2021. PMID: 34158028 Free PMC article. Review.
-
Anti-IL-5 therapies for chronic obstructive pulmonary disease.Cochrane Database Syst Rev. 2020 Dec 8;12(12):CD013432. doi: 10.1002/14651858.CD013432.pub2. Cochrane Database Syst Rev. 2020. PMID: 33295032 Free PMC article.
Cited by
-
Real-world evidence on biologic use in paediatric asthma: there but not there yet?ERJ Open Res. 2025 Jun 23;11(3):00209-2025. doi: 10.1183/23120541.00209-2025. eCollection 2025 May. ERJ Open Res. 2025. PMID: 40551792 Free PMC article.
-
An Updated Systematic Review on Asthma Exacerbation Risk Prediction Models Between 2017 and 2023: Risk of Bias and Applicability.J Asthma Allergy. 2025 Apr 19;18:579-589. doi: 10.2147/JAA.S509260. eCollection 2025. J Asthma Allergy. 2025. PMID: 40270986 Free PMC article. Review.
-
Epigenetic patient stratification via contrastive machine learning refines hallmark biomarkers in minoritized children with asthma.Res Sq [Preprint]. 2024 Sep 13:rs.3.rs-5066762. doi: 10.21203/rs.3.rs-5066762/v1. Res Sq. 2024. PMID: 39315258 Free PMC article. Preprint.
References
-
- Chung KF, Wenzel SE, Brozek JL, Bush A, Castro M, Sterk PJ, et al. International ERS/ATS guidelines on definition, evaluation and treatment of severe asthma. Eur Respir J. 2014 Feb;43(2):343–73. - PubMed
-
- Hekking PPW, Wener RR, Amelink M, Zwinderman AH, Bouvy ML, Bel EH. The prevalence of severe refractory asthma. J Allergy Clin Immunol. 2015 Apr;135(4):896–902. - PubMed
-
- von Bülow A, Kriegbaum M, Backer V, Porsbjerg C. The prevalence of severe asthma and low asthma control among danish adults. J Allergy Clin Immunol Pract. 2014 Dec;2(6):759–67. - PubMed
-
- Chen W, Safari A, FitzGerald JM, Sin DD, Tavakoli H, Sadatsafavi M. Economic burden of multimorbidity in patients with severe asthma: a 20-year population-based study. Thorax. 2019 Dec;74(12):1113–9. - PubMed

