Outcome and prognostic variables in childhood rhabdomyosarcoma (RMS) with emphasis on impact of FOXO1 fusions in non-metastatic RMS: experience from a tertiary cancer centre in India
- PMID: 37138963
- PMCID: PMC10151086
- DOI: 10.3332/ecancer.2023.1539
Outcome and prognostic variables in childhood rhabdomyosarcoma (RMS) with emphasis on impact of FOXO1 fusions in non-metastatic RMS: experience from a tertiary cancer centre in India
Abstract
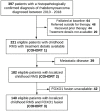
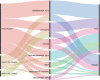
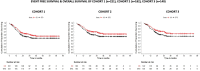
While factors influencing outcomes of rhabdomyosarcoma (RMS) in developed countries have evolved from clinical characteristics to molecular profiles, similar data from developing countries are scarce. This is a single-centre analysis of outcomes in treated cases of RMS, with emphasis on prevalence, risk-migration and prognostic impact of Forkhead Box O1 (FOXO1) in non-metastatic RMS. All children with histopathologically proven RMS, treated between January 2013 and December 2018 were included. Intergroup Rhabdomyosarcoma Study-4 risk stratification was used, with treatment based on a multimodality-regimen with chemotherapy (Vincristine/Ifosfamide/Etoposide and Vincristine/Actinomycin-D/Cyclophosphamide) and appropriate local therapy. Formalin-fixed paraffin-embedded tissues were tested using Reverse Transcriptase-Polymerase Chain Reaction for FOXO1-fusions (PAX3(P3F); PAX7(P7F)). A total of 221 children (Cohort-1) were included, of which 182 patients had non-metastatic disease (Cohort-2). Thirty-six (16%), 146 (66%), 39 (18%) patients were low-risk (LR), intermediate-risk (IR) and high-risk, respectively. FOXO1-fusion status was available in 140 patients with localised RMS (Cohort 3). P3F and P7F were detected in 25/49 (51%) and 14/85 (16.5%) of alveolar and embryonal variants, respectively. The 5-year-event-free survival (EFS)/overall survival (OS) of Cohorts 1, 2 and 3 was 48.5%/55.5%, 54.6%/62.6% and 55.1%/63.7%, respectively. Amongst the localised RMS, presence of nodal metastases and primary tumour size > 10 cms were adverse prognostic factorvs (p < 0.05). On incorporating fusion-status in risk-stratification, 6/29 (21%) patients migrated from LR (A/B) to IR. All patients who re-categorised as LR (FOXO1 negative) had a 5-year EFS/OS of 80.81%/90.91%. FOXO1-negative tumours had a better 5-year relapse-free survival (58.92% versus 44.63%; p = 0.296) with a near-significant correlation in favourable-site tumours (75.10% versus 45.83%; p = 0.063). While FOXO1-fusions have superior prognostic utility compared to histology alone in localised, favourable-site RMS, traditional prognostic factors (tumour size and nodal metastases) impacted outcome the most in this subset. Strengthening of early referral systems in community and timely local intervention can help in improving outcome in resource-constrained countries.
Keywords: FOXO1 fusions; India; LMIC; paediatric soft tissue sarcomas; rhabdomyosarcoma.
© the authors; licensee ecancermedicalscience.
Conflict of interest statement
The authors declare no conflicts of interest influencing the contents on this article.
Figures
References
-
- Newton WA, Gehan EA, Webber BL, et al. Classification of rhabdomyosarcomas and related sarcomas. Pathologic aspects and proposal for a new classification – an intergroup rhabdomyosarcoma study. Cancer. 1995;76(6):1073–1085. doi: 10.1002/1097-0142(19950915)76:6<1073::AID-CNCR2820760624>3.0.CO;2-L. - DOI - PubMed
-
- National Cancer Institute (NCI) A randomized phase 3 study of vincristine, dactinomycin, cyclophosphamide (VAC) alternating with vincristine and irinotecan (VI) versus VAC/VI plus temsirolimus (TORI, torisel, NSC# 683864) in patients with intermediate risk (IR) rhabdomyosarcoma (RMS) 2020. [clinicaltrials.gov]
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous