Interaction between myelodysplasia-related gene mutations and ontogeny in acute myeloid leukemia
- PMID: 37142255
- PMCID: PMC10471939
- DOI: 10.1182/bloodadvances.2023009675
Interaction between myelodysplasia-related gene mutations and ontogeny in acute myeloid leukemia
Abstract
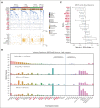
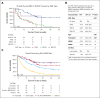
Accurate classification and risk stratification are critical for clinical decision making in patients with acute myeloid leukemia (AML). In the newly proposed World Health Organization and International Consensus classifications of hematolymphoid neoplasms, the presence of myelodysplasia-related (MR) gene mutations is included as 1 of the diagnostic criteria for AML, AML-MR, based largely on the assumption that these mutations are specific for AML with an antecedent myelodysplastic syndrome. ICC also prioritizes MR gene mutations over ontogeny (as defined in the clinical history). Furthermore, European LeukemiaNet (ELN) 2022 stratifies these MR gene mutations into the adverse-risk group. By thoroughly annotating a cohort of 344 newly diagnosed patients with AML treated at the Memorial Sloan Kettering Cancer Center, we show that ontogeny assignments based on the database registry lack accuracy. MR gene mutations are frequently observed in de novo AML. Among the MR gene mutations, only EZH2 and SF3B1 were associated with an inferior outcome in the univariate analysis. In a multivariate analysis, AML ontogeny had independent prognostic values even after adjusting for age, treatment, allo-transplant and genomic classes or ELN risks. Ontogeny also helped stratify the outcome of AML with MR gene mutations. Finally, de novo AML with MR gene mutations did not show an adverse outcome. In summary, our study emphasizes the importance of accurate ontogeny designation in clinical studies, demonstrates the independent prognostic value of AML ontogeny, and questions the current classification and risk stratification of AML with MR gene mutations.
© 2023 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Conflict of interest statement
Conflict-of-interest disclosure: M.E.A. served as a consultant for Janssen Global Services, Bristol Myers Squibb (BMS), AstraZeneca, and Roche, and has received honoraria from Biocartis, Invivoscribe, physician educational resources, PeerView Institute for Medical Education, clinical care options, and RMEI Medical Education. A.J.S. reports his spouse is an employee of BMS. B.J.B. served on the advisory board for Oncovalent and BMS. S.F.C. is a consultant for and holds an equity interest in Imago BioSciences, none of which is directly related to the content of this paper. J.L.G. received consulting fees from GLG. M.B.G. receives research support from Actinium, Amgen, and Sanofi, and has served in an advisory role for Sanofi, Novartis, and Allogene. R.K.R. has received consulting fees from Incyte Corporation, Celgene/BMS, Blueprint, AbbVie, CTI, Stemline, Galecto, Pharmaessentia, Constellation/MorphoSys, Sierra Oncology/GlaxoSmithKline, Sumitomo Dainippon, Kartos, Servier, Zentalis, Karyopharm, and research funding from Constellation Pharmaceuticals, Ryvu, Zentalis, and Stemline Therapeutics. O.I.A.-W. served as a consultant for H3 Biomedicine, Foundation Medicine Inc, Merck, Prelude Therapeutics, and Janssen; is on the scientific advisory board of Envisagenics Inc, AIChemy, Harmonic Discovery Inc, and Pfizer Boulder; and has received prior research funding from H3 Biomedicine, Nurix Therapeutics, Minovia Therapeutics, and Loxo Oncology, unrelated to the current manuscript. A.D.G. received research funding from Celularity, ADC Therapeutics, Aprea, AROG, Pfizer, Prelude, and Trillium; received research funding from and served as a consultant for Aptose and Daiichi Sankyo; served as a consultant and member of the advisory committees for Astellas, Celgene, and Genentech; received research funding from, served as a consultant for, and was a member of the advisory committees for AbbVie; and received honoraria from Dava Oncology. M.S.T. received research funding from AbbVie, Orsenix, BioSight, Glycomimetics, Rafael Pharmaceuticals, and Amgen; is on the advisory boards of AbbVie, Daiichi Sankyo, Orsenix, KAHR, Jazz Pharmaceuticals, Roche, BioSight, Novartis, Innate Pharmaceuticals, Kura, Syros Pharmaceuticals, and Ipsen Biopharmaceuticals; received royalties from UpToDate; and is on the DSMB of HOVON protocol Ho156 and adjudication committee of Foghorn protocol FHD-286. R.L.L. is on the supervisory board of Qiagen and is a scientific adviser to Imago, Mission Bio, Syndax, Zentalis, Ajax, Bakx, Auron, Prelude, C4 Therapeutics, and Isoplexis for which he receives equity support; receives research support from Ajax and AbbVie; has consulted for Incyte, Janssen, MorphoSys, and Novartis; and received honoraria from AstraZeneca and Kura for invited lectures and from Gilead for grant reviews. E.P. is a founder and equity holder, and holds a fiduciary role in Isabl Inc. M.R. is on the scientific advisory board in Auron Pharmaceutical for which he received equity support; receives research funding from Celularity, Roche-Genentech, Beat AML, and NGM; and receives travel funds from BD Biosciences. W.X. received research support from Stemline Therapeutics. The remaining authors declare no competing financial interests.
The current affiliation for Z.S.S.-M. is Jacobs School of Medicine and Biomedical Sciences at the University at Buffalo, Buffalo, NY.
The current affiliation for J.C. is Weill Cornell Medicine, New York City, NY.
The current affiliation for B.J.B. is City of Hope National Medical Center, Duarte, CA.
The current affiliation for M.S.T. is Northwestern University, Chicago, IL.
Figures



References
-
- Pollyea DA, Bixby D, Perl A, et al. NCCN guidelines insights: acute myeloid leukemia, version 2.2021. J Natl Compr Canc Netw. 2021;19(1):16–27. - PubMed
-
- Döhner H, Wei AH, Appelbaum FR, et al. Diagnosis and management of AML in adults: 2022 recommendations from an international expert panel on behalf of the ELN. Blood. 2022;140(12):1345–1377. - PubMed
-
- Bennett JM, Catovsky D, Daniel M-T, et al. Proposals for the classification of the Acute Leukaemias French-American-British (FAB) Co-operative Group. Br J Haematol. 1976;33(4):451–458. - PubMed
-
- Arber DA, Orazi A, Hasserjian R, et al. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016;127(20):2391–2405. - PubMed
-
- Vardiman JW, Thiele J, Arber DA, et al. The 2008 revision of the World Health Organization (WHO) classification of myeloid neoplasms and acute leukemia: rationale and important changes. Blood. 2009;114(5):937–951. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

