A History of Low-Dose Ethanol Shifts the Role of Ventral Hippocampus during Reward Seeking in Male Mice
- PMID: 37156611
- PMCID: PMC10198604
- DOI: 10.1523/ENEURO.0087-23.2023
A History of Low-Dose Ethanol Shifts the Role of Ventral Hippocampus during Reward Seeking in Male Mice
Abstract
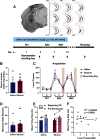
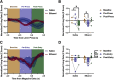
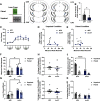
Although casual drinkers are a majority of the alcohol drinking population, understanding of the long-term effects of chronic exposure to lower levels of alcohol is limited. Chronic exposure to lower doses of ethanol may facilitate the development of alcohol use disorders, potentially because of ethanol effects on reward learning and motivation. Indeed, our previously published findings showed that chronic low-dose ethanol exposure enhanced motivation for sucrose in male, but not female, mice. As the ventral hippocampus (vHPC) is sensitive to disruption by higher doses of chronic ethanol and tracks reward-related information, we hypothesized that this region is impacted by low-dose ethanol and, further, that manipulating vHPC activity would alter reward motivation. In vivo electrophysiological recordings of vHPC population neural activity during progressive ratio testing revealed that vHPC activity was suppressed in the period immediately after reward seeking (lever press) in ethanol-naive controls, whereas suppression of vHPC activity anticipated reward seeking in ethanol-exposed mice. In both ethanol-naive and exposed mice, vHPC activity was suppressed before a reward magazine entry. Temporally selective inhibition of vHPC using optogenetics increased motivation for sucrose in ethanol-naive controls, but not in ethanol-exposed mice. Further, regardless of exposure history, vHPC inhibition promoted checking of the reward magazine, indicating a role for vHPC in reward tracking. There was no effect of chemogenetic inhibition of the vHPC either during training or testing on sucrose reward motivation. These results reveal novel ethanol-induced alterations in vHPC neural activity that shift how vHPC activity is able to regulate reward seeking.
Keywords: electrophysiology; ethanol; hippocampus; motivation; operant; optogenetics.
Copyright © 2023 Bryant et al.
Conflict of interest statement
The authors declare no competing financial interests.
Figures
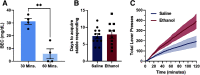
Similar articles
-
Reinforcement History Dependent Effects of Low Dose Ethanol on Reward Motivation in Male and Female Mice.Front Behav Neurosci. 2022 Apr 11;16:875890. doi: 10.3389/fnbeh.2022.875890. eCollection 2022. Front Behav Neurosci. 2022. PMID: 35481242 Free PMC article.
-
Effects of chronic alcohol exposure on motivation-based value updating.Alcohol. 2022 Jun;101:53-64. doi: 10.1016/j.alcohol.2022.03.004. Epub 2022 Apr 8. Alcohol. 2022. PMID: 35405249 Free PMC article.
-
Ethanol-associated cues produce general pavlovian-instrumental transfer.Alcohol Clin Exp Res. 2007 May;31(5):766-74. doi: 10.1111/j.1530-0277.2007.00359.x. Epub 2007 Mar 22. Alcohol Clin Exp Res. 2007. PMID: 17378919
-
[Epigenetic mechanisms and alcohol use disorders: a potential therapeutic target].Biol Aujourdhui. 2017;211(1):83-91. doi: 10.1051/jbio/2017014. Epub 2017 Jul 6. Biol Aujourdhui. 2017. PMID: 28682229 Review. French.
-
Linking external stimuli with internal drives: A role for the ventral hippocampus.Curr Opin Neurobiol. 2022 Oct;76:102590. doi: 10.1016/j.conb.2022.102590. Epub 2022 Jun 23. Curr Opin Neurobiol. 2022. PMID: 35753108 Free PMC article. Review.
Cited by
-
Sex and individual differences in the effect of chronic low-dose ethanol on behavioral strategy selection.Alcohol Clin Exp Res (Hoboken). 2024 Jan;48(1):132-141. doi: 10.1111/acer.15218. Epub 2023 Nov 8. Alcohol Clin Exp Res (Hoboken). 2024. PMID: 38206280 Free PMC article.
-
Dissociating the incubation of appetitive and consummatory behavior in a model of oral cocaine self-administration.bioRxiv [Preprint]. 2025 Feb 13:2025.02.12.637922. doi: 10.1101/2025.02.12.637922. bioRxiv. 2025. Update in: Pharmacol Biochem Behav. 2025 Sep;254:174055. doi: 10.1016/j.pbb.2025.174055. PMID: 39990380 Free PMC article. Updated. Preprint.
-
Historical loss weakens competitive behavior by remodeling ventral hippocampal dynamics.Cell Discov. 2025 Feb 25;11(1):16. doi: 10.1038/s41421-024-00751-3. Cell Discov. 2025. PMID: 39994206 Free PMC article.
References
-
- Bach EC, Morgan JW, Ewin SE, Barth SH, Raab-Graham KF, Weiner JL (2021) Chronic ethanol exposures leads to a negative affective state in female rats that is accompanied by a paradoxical decrease in ventral hippocampus excitability. Front Neurosci 15:669075. 10.3389/fnins.2021.669075 - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
