Neurotrophins and Their Receptors, Novel Therapeutic Targets for Pelvic Pain in Endometriosis, Are Coordinately Regulated by IL-1β via the JNK Signaling Pathway
- PMID: 37164275
- PMCID: PMC10433690
- DOI: 10.1016/j.ajpath.2023.04.007
Neurotrophins and Their Receptors, Novel Therapeutic Targets for Pelvic Pain in Endometriosis, Are Coordinately Regulated by IL-1β via the JNK Signaling Pathway
Abstract
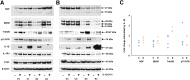
Pelvic pain in women with endometriosis is attributed to neuroinflammation and afferent nociceptor nerves in ectopic and eutopic endometrium. The hypothesis that uterine nociception is activated by IL-1β, a prominent cytokine in endometriosis, was tested herein. Immunofluorescence histochemistry confirmed the presence of neurons in human endometrial tissue. Expression of nerve growth factor (NGF) and brain-derived neurotrophic factor (BDNF) and their receptors in endometrial tissue and cells was validated by immunohistochemistry and Western blotting. Isolated endometrial stromal cells (ESCs) were subjected to dose-response and time-course experiments with IL-1β and kinase inhibitors to characterize in vitro biomarkers. Neural biomarkers were co-localized in endometrial nerve fibers. NGF, BDNF, and their receptors tropomyosin receptor kinase (Trk) A, TrkB, and p75 neurotrophin receptor were all expressed in primary ESCs. IL-1β stimulated higher TrkA/B expression in ESCs derived from endometriosis cases (2.8- ± 0.2-fold) than cells from controls (1.5- ± 0.3-fold, t-test, P < 0.01), effects that were mediated via the c-Jun N-terminal kinase (JNK) pathway. BDNF concentrations trended higher in peritoneal fluid of endometriosis cases but were not statistically different from controls (P = 0.16). The results support the hypothesis that NGF and BDNF and their corresponding receptors orchestrate innervation of the endometrium, which is augmented by IL-1β. We postulate that JNK inhibitors, such as SP600125, have the potential to reduce neuroinflammation in women with endometriosis.
Copyright © 2023 American Society for Investigative Pathology. Published by Elsevier Inc. All rights reserved.
Figures





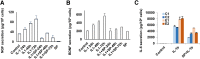
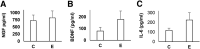

References
-
- As-Sanie S., Black R., Giudice L.C., Gray Valbrun T., Gupta J., Jones B., Laufer M.R., Milspaw A.T., Missmer S.A., Norman A., Taylor R.N., Wallace K., Williams Z., Yong P.J., Nebel R.A. Assessing research gaps and unmet needs in endometriosis. Am J Obstet Gynecol. 2019;221:86–94. - PubMed
-
- Davenport A.J., Neagoe I., Bräuer N., Koch M., Rotgeri A., Nagel J., Laux-Biehlmann A., Machet F., Coelho A.M., Boyce S., Carty N., Gemkow M.J., Hess S.D., Zollner T.M., Fischer O.M. Eliapixant is a selective P2X3 receptor antagonist for the treatment of disorders associated with hypersensitive nerve fibers. Sci Rep. 2021;11 - PMC - PubMed
-
- Asante A., Taylor R.N. Endometriosis: the role of neuroangiogenesis. Annu Rev Physiol. 2011;73:163–182. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

