Targeting pyroptosis as a preventive and therapeutic approach for stroke
- PMID: 37165005
- PMCID: PMC10172388
- DOI: 10.1038/s41420-023-01440-y
Targeting pyroptosis as a preventive and therapeutic approach for stroke
Abstract
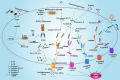
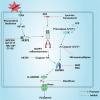
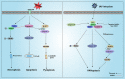
Stroke has caused tremendous social stress worldwide, yet despite decades of research and development of new stroke drugs, most have failed and rt-PA (Recombinant tissue plasminogen activator) is still the accepted treatment for ischemic stroke. the complexity of the stroke mechanism has led to unsatisfactory efficacy of most drugs in clinical trials, indicating that there are still many gaps in our understanding of stroke. Pyroptosis is a programmed cell death (PCD) with inflammatory properties and are thought to be closely associated with stroke. Pyroptosis is regulated by the GSDMD of the gasdermin family, which when cleaved by Caspase-1/Caspase-11 into N-GSDMD with pore-forming activity can bind to the plasma membrane to form small 10-20 nm pores, which would allow the release of inflammatory factors IL-18 and IL-1β before cell rupture, greatly exacerbating the inflammatory response. The pyroptosis occurs mainly in the border zone of cerebral infarction, and glial cells, neuronal cells and brain microvascular endothelial cells (BMECs) all undergo pyroptosis after stroke, which largely exacerbates the breakdown of the blood-brain barrier (BBB) and thus aggravates brain injury. Therefore, pyroptosis may be a good direction for the treatment of stroke. In this review, we focus on the latest mechanisms of action of pyroptosis and the process by which pyroptosis regulates stroke development. We also suggest potential therapeutic stroke drugs that target the pyroptosis pathway, providing additional therapeutic strategies for the clinical management of stroke. The role of pyroptosis after stroke. After stroke, microglia first rush to the damaged area and polarize into M1 and M2 types. Under the influence of various stimuli, microglia undergo pyroptosis, release pro-inflammatory factors, and are converted to the M1 type; astrocytes and neuronal cells also undergo pyroptosis under the stimulation of various pro-inflammatory factors, leading to astrocyte death due to increased osmotic pressure in the membrane, resulting in water absorption and swelling until rupture. BMECs, the main structural component of the BBB, also undergo pyroptosis when stimulated by pro-inflammatory factors released from microglia and astrocytes, leading to the destruction of the structural integrity of the BBB, ultimately causing more severe brain damage. In addition, GSDMD in neutrophils mainly mediate the release of NETs rather than pyroptosis, which also aggravates brain injury. IL-10=interleukin-10; TGF-β = transforming growth factor-β; IL-18=interleukin-18; IL-1β = interleukin-1β; TNF-α = tumor necrosis factor-α; iNOS=induced nitrogen monoxide synthase; MMPs=Matrix metalloproteinases; GSDMD = gasdermin D; BMECs=brain microvascular endothelial cells; BBB = blood-brain barrier.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


References
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

