One assay to test them all: Multiplex assays for expansion of respiratory virus surveillance
- PMID: 37168265
- PMCID: PMC10165998
- DOI: 10.3389/fmed.2023.1161268
One assay to test them all: Multiplex assays for expansion of respiratory virus surveillance
Abstract
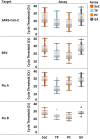
Molecular multiplex assays (MPAs) for simultaneous detection of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), influenza and respiratory syncytial virus (RSV) in a single RT-PCR reaction reduce time and increase efficiency to identify multiple pathogens with overlapping clinical presentation but different treatments or public health implications. Clinical performance of XpertXpress® SARS-CoV-2/Flu/RSV (Cepheid, GX), TaqPath™ COVID-19, FluA/B, RSV Combo kit (Thermo Fisher Scientific, TP), and PowerChek™ SARS-CoV-2/Influenza A&B/RSV Multiplex RT-PCR kit II (KogeneBiotech, PC) was compared to individual Standards of Care (SoC). Thirteen isolates of SARS-CoV-2, human seasonal influenza, and avian influenza served to assess limit of detection (LoD). Then, positive and negative residual nasopharyngeal specimens, collected under public health surveillance and pandemic response served for evaluation. Subsequently, comparison of effectiveness was assessed. The three MPAs confidently detect all lineages of SARS-CoV-2 and influenza viruses. MPA-LoDs vary from 1 to 2 Log10 differences from SoC depending on assay and strain. Clinical evaluation resulted in overall agreement between 97 and 100%, demonstrating a high accuracy to detect all targets. Existing differences in costs, testing burden and implementation constraints influence the choice in primary or community settings. TP, PC and GX, reliably detect SARS-CoV-2, influenza and RSV simultaneously, with reduced time-to-results and simplified workflows. MPAs have the potential to enhance diagnostics, surveillance system, and epidemic response to drive policy on prevention and control of viral respiratory infections.
Keywords: COVID-19; SARS-CoV-2; diagnostic test; influenza; multiplex assay; respiratory syncytial virus; surveillance.
Copyright © 2023 Boukli, Flamand, Chea, Heng, Keo, Sour, In, Chhim, Chhor, Kruy, Feenstra, Gandhi, Okafor, Ulekleiv, Auerswald, Horm and Karlsson.
Conflict of interest statement
JF, MG, OO, and CU were employed by Thermo Fisher Scientific. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Update of
-
ONE ASSAY TO TEST THEM ALL: COMPARING MULTIPLEX ASSAYS FOR EXPANSION OF RESPIRATORY VIRUS SURVEILLANCE.medRxiv [Preprint]. 2023 Jan 22:2023.01.19.23284806. doi: 10.1101/2023.01.19.23284806. medRxiv. 2023. Update in: Front Med (Lausanne). 2023 Apr 24;10:1161268. doi: 10.3389/fmed.2023.1161268. PMID: 36711477 Free PMC article. Updated. Preprint.
References
-
- World Health Organization . WHO the Top 10 Causes of Death. (2020). Available at: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (Accessed August 12, 2022).
-
- Perin J, Mulick A, Yeung D, Villavicencio F, Lopez G, Strong KL, et al. . Global, regional, and national causes of under-5 mortality in 2000–19: an updated systematic analysis with implications for the sustainable development goals. Lancet Child Adolesc Health. (2022) 6:106–15. doi: 10.1016/S2352-4642(21)00311-4, PMID: - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

