ERBB2 Amplification as a Predictive and Prognostic Biomarker in Upper Tract Urothelial Carcinoma
- PMID: 37173881
- PMCID: PMC10177383
- DOI: 10.3390/cancers15092414
ERBB2 Amplification as a Predictive and Prognostic Biomarker in Upper Tract Urothelial Carcinoma
Abstract
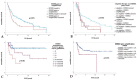
Upper tract urothelial carcinomas (UTUCs) occur in about 5-10% of all urothelial carcinomas and are frequently discovered in high-stage disease. We aimed to evaluate human epidermal growth factor receptor 2 (ERBB2) protein expression immunohistochemically and ERBB2 amplification in UTUCs by fluorescence in situ hybridization, applying a tissue microarray technique. ERBB2 overexpression and ERBB2 amplification were defined according to the recommendations of the American Society of Clinical Oncology/College of American Pathologists (ASCO/CAP) for breast cancer and gastric carcinoma (GC), revealing scores of 2+ and 3+ in 10.2% and 41.8% of UTUCs, respectively. The performance parameters showed obviously higher sensitivity of ERBB2 immunoscoring according to the ASCO/CAP criteria for GC. ERBB2 amplification was detected in 10.5% of UTUCs. ERBB2 overexpression was more likely to be found in high-grade tumors and was associated with tumor progression. Univariable Cox regression analysis revealed a significantly lower progression-free survival (PFS) in cases with ERBB2 immunoscores of 2+ or 3+ according to the ASCO/CAP guidelines for GC. UTUCs with ERBB2 amplification showed a significantly shorter PFS in the multivariable Cox regression analysis. Irrespective of their ERBB2 status, patients with UTUC treated with platin showed a significantly lower PFS than UTUC patients who had not received any platin-based therapy. In addition, UTUC patients with a normal ERBB2 gene status who had not received platin-based therapy showed significantly longer overall survival. The results suggest that ERBB2 is a biomarker for progression in UTUCs and may define a distinct subgroup of UTUCs. As previously shown, ERBB2 amplification is infrequent. However, the small number of patients diagnosed with ERBB2-amplified UTUC might benefit from ERBB2-targeted cancer therapy. In clinical-pathological routine diagnostics, the determination of ERBB2 amplification is an established method in some defined entities and also successful in small samples. Still, the simultaneous use of ERBB2 immunohistochemistry and ERBB2 in situ hybridization would be important in order to record the low rate of amplified UTUC cases as completely as possible.
Keywords: ERBB2; fluorescence in situ hybridization; survival analysis; tissue microarray; upper tract urothelial carcinoma.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures

Similar articles
-
Next-generation assessment of human epidermal growth factor receptor 2 gene (ERBB2) amplification status in invasive breast carcinoma: a focus on Group 4 by use of the 2018 American Society of Clinical Oncology/College of American Pathologists HER2 testing guideline.Histopathology. 2021 Mar;78(4):498-507. doi: 10.1111/his.14241. Epub 2020 Oct 12. Histopathology. 2021. PMID: 32841416 Free PMC article.
-
ERBB2 Amplification and HER2 Expression in Salivary Duct Carcinoma: Evaluation of Scoring Guidelines and Potential for Expanded Anti-HER2 Therapy.Mod Pathol. 2023 Oct;36(10):100273. doi: 10.1016/j.modpat.2023.100273. Epub 2023 Jul 7. Mod Pathol. 2023. PMID: 37423585
-
Outcome of patients with micropapillary urothelial carcinoma following radical cystectomy: ERBB2 (HER2) amplification identifies patients with poor outcome.Mod Pathol. 2014 May;27(5):758-64. doi: 10.1038/modpathol.2013.201. Epub 2013 Nov 1. Mod Pathol. 2014. PMID: 24186136
-
Accuracy and Clinical Utility of a Tumor Grade- and Stage-based Predictive Model in Localized Upper Tract Urothelial Carcinoma.Eur Urol Focus. 2022 May;8(3):761-768. doi: 10.1016/j.euf.2021.05.002. Epub 2021 May 27. Eur Urol Focus. 2022. PMID: 34053904 Review.
-
Upper tract urothelial carcinoma: a different disease entity in terms of management.ESMO Open. 2017 Jan 24;1(6):e000126. doi: 10.1136/esmoopen-2016-000126. eCollection 2016. ESMO Open. 2017. PMID: 28848663 Free PMC article. Review.
Cited by
-
Current and Emerging Strategies to Treat Urothelial Carcinoma.Cancers (Basel). 2023 Oct 8;15(19):4886. doi: 10.3390/cancers15194886. Cancers (Basel). 2023. PMID: 37835580 Free PMC article. Review.
-
Lymph Node Dissection in Upper Tract Urothelial Carcinoma: Current Status and Future Perspectives.Curr Oncol Rep. 2023 Nov;25(11):1327-1344. doi: 10.1007/s11912-023-01460-y. Epub 2023 Oct 6. Curr Oncol Rep. 2023. PMID: 37801187 Free PMC article. Review.
-
Near Complete Response to Platinum-based Systemic Chemotherapy in High-risk Upper Tract Urothelial Carcinoma With an ERBB2 Gene Mutation: A Case Report.Urology. 2024 Feb;184:75-78. doi: 10.1016/j.urology.2023.11.019. Epub 2023 Dec 3. Urology. 2024. PMID: 38052324 Free PMC article.
References
-
- Rouprêt M., Babjuk M., Burger M., Capoun O., Cohen D., Compérat E.M., Cowan N.C., Dominguez-Escrig J.L., Gontero P., Hugh Mostafid A., et al. European Association of Urology Guidelines on Upper Urinary Tract Urothelial Carcinoma: 2020 Update. Eur. Urol. 2020;79:62–79. doi: 10.1016/j.eururo.2020.05.042. - DOI - PubMed
-
- Rouprêt M., Babjuk M., Compérat E., Zigeuner R., Sylvester R.J., Burger M., Cowan N.C., Böhle A., van Rhijn B.W.G., Kaasinen E., et al. European Association of Urology Guidelines on Upper Urinary Tract Urothelial Cell Carcinoma: 2015 Update. Eur. Urol. 2015;68:868–879. doi: 10.1016/j.eururo.2015.06.044. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

