Phosphoproteomic Approaches for Identifying Phosphatase and Kinase Substrates
- PMID: 37175085
- PMCID: PMC10180314
- DOI: 10.3390/molecules28093675
Phosphoproteomic Approaches for Identifying Phosphatase and Kinase Substrates
Abstract
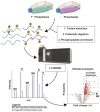
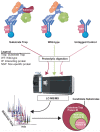
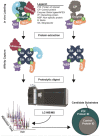
Protein phosphorylation is a ubiquitous post-translational modification controlled by the opposing activities of protein kinases and phosphatases, which regulate diverse biological processes in all kingdoms of life. One of the key challenges to a complete understanding of phosphoregulatory networks is the unambiguous identification of kinase and phosphatase substrates. Liquid chromatography-coupled mass spectrometry (LC-MS/MS) and associated phosphoproteomic tools enable global surveys of phosphoproteome changes in response to signaling events or perturbation of phosphoregulatory network components. Despite the power of LC-MS/MS, it is still challenging to directly link kinases and phosphatases to specific substrate phosphorylation sites in many experiments. Here, we survey common LC-MS/MS-based phosphoproteomic workflows for identifying protein kinase and phosphatase substrates, noting key advantages and limitations of each. We conclude by discussing the value of inducible degradation technologies coupled with phosphoproteomics as a new approach that overcomes some limitations of current methods for substrate identification of kinases, phosphatases, and other regulatory enzymes.
Keywords: PTM; kinase; mass spectrometry; phosphatase; phosphoproteomics; phosphorylation; post-translation modification; quantitative proteomics; substrate identification.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

References
-
- Mertins P., Tang L.C., Krug K., Clark D.J., Gritsenko M.A., Chen L., Clauser K.R., Clauss T.R., Shah P., Gillette M.A., et al. Reproducible workflow for multiplexed deep-scale proteome and phosphoproteome analysis of tumor tissues by liquid chromatography–mass spectrometry. Nat. Protoc. 2018;13:1632–1661. doi: 10.1038/s41596-018-0006-9. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

