Distinct Profiles of Desensitization of µ-Opioid Receptors Caused by Remifentanil or Fentanyl: In Vitro Assay with Cells and Three-Dimensional Structural Analyses
- PMID: 37176075
- PMCID: PMC10179353
- DOI: 10.3390/ijms24098369
Distinct Profiles of Desensitization of µ-Opioid Receptors Caused by Remifentanil or Fentanyl: In Vitro Assay with Cells and Three-Dimensional Structural Analyses
Abstract
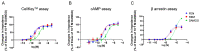
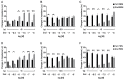
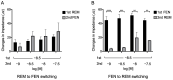
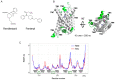
Remifentanil (REM) and fentanyl (FEN) are commonly used analgesics that act by activating a µ-opioid receptor (MOR). Although optimal concentrations of REM can be easily maintained during surgery, it is sometimes switched to FEN for optimal pain regulation. However, standards for this switching protocol remain unclear. Opioid anesthetic efficacy is decided in part by MOR desensitization; thus, in this study, we investigated the desensitization profiles of REM and FEN to MOR. The efficacy and potency during the 1st administration of REM or FEN in activating the MOR were almost equal. Similarly, in β arrestin recruitment, which determines desensitization processes, they showed no significant differences. In contrast, the 2nd administration of FEN resulted in a stronger MOR desensitization potency than that of REM, whereas REM showed a higher internalization potency than FEN. These results suggest that different β arrestin-mediated signaling caused by FEN or REM led to their distinct desensitization and internalization processes. Our three-dimensional analysis, with in silico binding of REM and FEN to MOR models, highlighted that REM and FEN bound to similar but distinct sites of MOR and led to distinct β arrestin-mediated profiles, suggesting that distinct binding profiles to MOR may alter β arrestin activity, which accounts for MOR desensitization and internalization.
Keywords: desensitization; fentanyl; in silico simulation; opioid receptor; remifentanil.
Conflict of interest statement
One author (K.O.) is an employee of a pharmaceutical company (Tsumura and Co.). However, the present study had no financial relationships with this company, as it was entirely sponsored by and performed at the Jikei University School of Medicine and Juntendo University Graduate School of Medicine.
Figures


Similar articles
-
Ketamine Improves Desensitization of µ-Opioid Receptors Induced by Repeated Treatment with Fentanyl but Not with Morphine.Biomolecules. 2022 Mar 10;12(3):426. doi: 10.3390/biom12030426. Biomolecules. 2022. PMID: 35327617 Free PMC article.
-
Usefulness for the combination of G-protein- and β-arrestin-biased ligands of μ-opioid receptors: Prevention of antinociceptive tolerance.Mol Pain. 2017 Jan-Dec;13:1744806917740030. doi: 10.1177/1744806917740030. Mol Pain. 2017. PMID: 29056067 Free PMC article.
-
Spinal Opioid Tolerance Depends upon Platelet-Derived Growth Factor Receptor-β Signaling, Not μ-Opioid Receptor Internalization.Mol Pharmacol. 2020 Oct;98(4):487-496. doi: 10.1124/mol.120.119552. Epub 2020 Jul 28. Mol Pharmacol. 2020. PMID: 32723769 Free PMC article.
-
Mechanisms of rapid opioid receptor desensitization, resensitization and tolerance in brain neurons.Br J Pharmacol. 2012 Mar;165(6):1704-1716. doi: 10.1111/j.1476-5381.2011.01482.x. Br J Pharmacol. 2012. PMID: 21564086 Free PMC article. Review.
-
The role of opioid receptor internalization and beta-arrestins in the development of opioid tolerance.Anesth Analg. 2005 Sep;101(3):728-734. doi: 10.1213/01.ANE.0000160588.32007.AD. Anesth Analg. 2005. PMID: 16115983 Review.
Cited by
-
Effects of different doses of remifentanil combined with sevoflurane anesthesia on postoperative analgesia and hemodynamics in pediatric patients undergoing laparoscopic inguinal hernia repair.BMC Anesthesiol. 2025 May 15;25(1):244. doi: 10.1186/s12871-025-03104-z. BMC Anesthesiol. 2025. PMID: 40375067 Free PMC article. Clinical Trial.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

