Exogenous tetracosahexaenoic acid modifies the fatty acid composition of human primary T lymphocytes and Jurkat T cell leukemia cells contingent on cell type
- PMID: 37177900
- PMCID: PMC10946481
- DOI: 10.1002/lipd.12372
Exogenous tetracosahexaenoic acid modifies the fatty acid composition of human primary T lymphocytes and Jurkat T cell leukemia cells contingent on cell type
Abstract
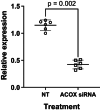
Tetracosahexaenoic acid (24:6ω-3) is an intermediate in the conversion of 18:3ω-3 to 22:6ω-3 in mammals. There is limited information about whether cells can assimilate and metabolize exogenous 24:6ω-3. This study compared the effect of incubation with 24:6ω-3 on the fatty acid composition of two related cell types, primary CD3+ T lymphocytes and Jurkat T cell leukemia, which differ in the integrity of the polyunsaturated fatty acid (PUFA) biosynthesis pathway. 24:6ω-3 was only detected in either cell type when cells were incubated with 24:6ω-3. Incubation with 24:6ω-3 induced similar increments in the amount of 22:6ω-3 in both cell types and modified the homeoviscous adaptations fatty acid composition induced by activation of T lymphocytes. The effect of incubation with 18:3ω-3 compared to 24:6ω-3 on the increment in 22:6ω-3 was tested in Jurkat cells because primary T cells cannot convert 18:3ω-3 to 22:6ω-3. The increment in the 22:6ω-3 content of Jurkat cells incubated with 24:6ω-3 was 19.5-fold greater than that of cells incubated with 18:3ω-3. Acyl-coA oxidase siRNA knockdown decreased the amount of 22:6ω-3 and increased the amount of 24:6ω-3 in Jurkat cells. These findings show exogenous 24:6ω-3 can be incorporated into primary human T lymphocytes and Jurkat cells and induces changes in fatty acid composition consistent with its conversion to 22:6ω-3 via a mechanism involving peroxisomal β-oxidation that is regulated independently from the integrity of the upstream PUFA synthesis pathway. One further implication is that consuming 24:6ω-3 may be an effective alternative means of achieving health benefits attributed to 20:5ω-3 and 22:6ω-3.
Keywords: desaturases; docosahexaenoic acid; fatty acid metabolism; general area; immunology; lipid biochemistry; mammalian lipid biochemistry; metabolism; n-3 fatty acids; polyunsaturated fatty acids (PUFA); specific lipids.
© 2023 The Authors. Lipids published by Wiley Periodicals LLC on behalf of AOCS.
Conflict of interest statement
The authors declare that they have no conflict of interest.
Figures


References
-
- Abraham RT, Weiss A. Jurkat T cells and development of the T‐cell receptor signalling paradigm. Nat Rev Immunol. 2004;4:301–8. - PubMed
-
- Anel A, Naval J, Gonzalez B, Torres JM, Mishal Z, Uriel J, et al. Fatty acid metabolism in human lymphocytes. I. Time‐course changes in fatty acid composition and membrane fluidity during blastic transformation of peripheral blood lymphocytes. Biochim Biophys Acta. 1990;1044:323–31. - PubMed
-
- Baes M, Gressens P, Baumgart E, Carmeliet P, Casteels M, Fransen M, et al. A mouse model for Zellweger syndrome. Nat Genet. 1997;17:49–57. - PubMed
-
- Bligh EG, Dyer WJ. A rapid method of total lipid extraction and purification. Can J Biochem Physiol. 1959;37:911–7. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

