Association of dynamic contrast-enhanced MRI and 18F-Fluorodeoxyglucose PET/CT parameters with neoadjuvant therapy response and survival in esophagogastric cancer
- PMID: 37183047
- PMCID: PMC10769883
- DOI: 10.1016/j.ejso.2023.05.009
Association of dynamic contrast-enhanced MRI and 18F-Fluorodeoxyglucose PET/CT parameters with neoadjuvant therapy response and survival in esophagogastric cancer
Abstract
Introduction: Better predictive markers are needed to deliver individualized care for patients with primary esophagogastric cancer. This exploratory study aimed to assess whether pre-treatment imaging parameters from dynamic contrast-enhanced MRI and 18F-fluorodeoxyglucose (18F-FDG) PET/CT are associated with response to neoadjuvant therapy or outcome.
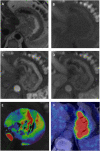
Materials and methods: Following ethical approval and informed consent, prospective participants underwent dynamic contrast-enhanced MRI and 18F-FDG PET/CT prior to neoadjuvant chemotherapy/chemoradiotherapy ± surgery. Vascular dynamic contrast-enhanced MRI and metabolic 18F-FDG PET parameters were compared by tumor characteristics using Mann Whitney U test and with pathological response (Mandard tumor regression grade), recurrence-free and overall survival using logistic regression modelling, adjusting for predefined clinical variables.
Results: 39 of 47 recruited participants (30 males; median age 65 years, IQR: 54, 72 years) were included in the final analysis. The tumor vascular-metabolic ratio was higher in patients remaining node positive following neoadjuvant therapy (median tumor peak enhancement/SUVmax ratio: 0.052 vs. 0.023, p = 0.02). In multivariable analysis adjusted for age, gender, pre-treatment tumor and nodal stage, peak enhancement (highest gadolinium concentration value prior to contrast washout) was associated with pathological tumor regression grade. The odds of response decreased by 5% for each 0.01 unit increase (OR 0.95; 95% CI: 0.90, 1.00, p = 0.04). No 18F-FDG PET/CT parameters were predictive of pathological tumor response. No relationships between pre-treatment imaging and survival were identified.
Conclusion: Pre-treatment esophagogastric tumor vascular and metabolic parameters may provide additional information in assessing response to neoadjuvant therapy.
Keywords: Esophageal cancer; Fluorodexoyglucose F18; Magnetic resonance imaging; Perfusion imaging; Positron emission tomography computed tomography.
Copyright © 2023 The Authors. Published by Elsevier Ltd.. All rights reserved.
Figures


References
-
- Edgren G., et al. A global assessment of the oesophageal adenocarcinoma epidemic. Gut. 2013;62(10):1406–1414. - PubMed
-
- Lordick F., et al. PET to assess early metabolic response and to guide treatment of adenocarcinoma of the oesophagogastric junction: the MUNICON phase II trial. Lancet Oncol. 2007;8(9):797–805. - PubMed
-
- Sjoquist K.M., et al. Survival after neoadjuvant chemotherapy or chemoradiotherapy for resectable oesophageal carcinoma: an updated meta-analysis. Lancet Oncol. 2011;12(7):681–692. - PubMed
-
- Lagergren P., et al. Health-related quality of life among patients cured by surgery for esophageal cancer. Cancer. 2007;110(3):686–693. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

