Expectation violations produce error signals in mouse V1
- PMID: 37183176
- PMCID: PMC10321125
- DOI: 10.1093/cercor/bhad163
Expectation violations produce error signals in mouse V1
Abstract
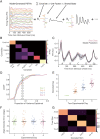
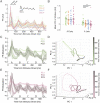
Repeated exposure to visual sequences changes the form of evoked activity in the primary visual cortex (V1). Predictive coding theory provides a potential explanation for this, namely that plasticity shapes cortical circuits to encode spatiotemporal predictions and that subsequent responses are modulated by the degree to which actual inputs match these expectations. Here we use a recently developed statistical modeling technique called Model-Based Targeted Dimensionality Reduction (MbTDR) to study visually evoked dynamics in mouse V1 in the context of an experimental paradigm called "sequence learning." We report that evoked spiking activity changed significantly with training, in a manner generally consistent with the predictive coding framework. Neural responses to expected stimuli were suppressed in a late window (100-150 ms) after stimulus onset following training, whereas responses to novel stimuli were not. Substituting a novel stimulus for a familiar one led to increases in firing that persisted for at least 300 ms. Omitting predictable stimuli in trained animals also led to increased firing at the expected time of stimulus onset. Finally, we show that spiking data can be used to accurately decode time within the sequence. Our findings are consistent with the idea that plasticity in early visual circuits is involved in coding spatiotemporal information.
Keywords: plasticity; prediction errors; primary visual cortex; statistical modeling.
© The Author(s) 2023. Published by Oxford University Press. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Figures




References
-
- Aoi MC, Pillow JW. Model-Based Targeted Dimensionality Reduction for neuronal population data. Neural Inf Process Syst. 2018:31:6690–6699. Retrieved from http://pillowlab.princeton.edu/jpillow/. - PMC - PubMed
-
- Atick JJ, Redlich AN. Convergent algorithm for sensory receptive field development. Neural Comput. 1993:5(1):45–60. 10.1162/neco.1993.5.1.45. - DOI
-
- Barlow HB. Possible principles underlying the transformations of sensory messages. In Sens Commun. 1961:1:217–234. 10.7551/mitpress/9780262518420.003.0013. - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

