Surfactant-Assisted Label-Free Fluorescent Aptamer Biosensors and Binding Assays
- PMID: 37185509
- PMCID: PMC10135756
- DOI: 10.3390/bios13040434
Surfactant-Assisted Label-Free Fluorescent Aptamer Biosensors and Binding Assays
Abstract
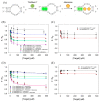
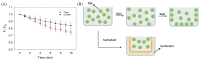
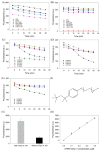
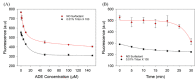
Using DNA staining dyes such as SYBR Green I (SGI) and thioflavin T (ThT) to perform label-free detection of aptamer binding has been performed for a long time for both binding assays and biosensor development. Since these dyes are cationic, they can also adsorb to the wall of reaction vessels leading to unstable signals and even false interpretations of the results. In this work, the stability of the signal was first evaluated using ThT and the classic adenosine aptamer. In a polystyrene microplate, a drop in fluorescence was observed even when non-binding targets or water were added, whereas a more stable signal was achieved in a quartz cuvette. Equilibrating the system can also improve signal stability. In addition, a few polymers and surfactants were also screened, and 0.01% Triton X-100 was found to have the best protection effect against fluorescence signal decrease due to dye adsorption. Three aptamers for Hg2+, adenosine, and cortisol were tested for their sensitivity and signal stability in the absence and presence of Triton X-100. In each case, the sensitivity was similar, whereas the signal stability was better for the surfactant. This study indicates that careful control experiments need to be designed to ensure reliable results and that the reliability can be improved by using Triton X-100 and a long equilibration time.
Keywords: aptamers; biosensors; fluorescence; surfactant.
Conflict of interest statement
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
Figures


References
-
- Daems E., Moro G., Campos R., De Wael K. Mapping the gaps in chemical analysis for the characterisation of aptamer-target interactions. TrAC Trends Anal. Chem. 2021;142:116311. doi: 10.1016/j.trac.2021.116311. - DOI
-
- McKeague M., De Girolamo A., Valenzano S., Pascale M., Ruscito A., Velu R., Frost N.R., Hill K., Smith M., McConnell E.M., et al. Comprehensive Analytical Comparison of Strategies Used for Small Molecule Aptamer Evaluation. Anal. Chem. 2015;87:8608–8612. doi: 10.1021/acs.analchem.5b02102. - DOI - PubMed
-
- He L., Huang R., Xiao P., Liu Y., Jin L., Liu H., Li S., Deng Y., Chen Z., Li Z., et al. Current signal amplification strategies in aptamer-based electrochemical biosensor: A review. Chin. Chem. Lett. 2021;32:1593–1602. doi: 10.1016/j.cclet.2020.12.054. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

