Establishment of Skeletal Myogenic Progenitors from Non-Human Primate Induced Pluripotent Stem Cells
- PMID: 37190056
- PMCID: PMC10137227
- DOI: 10.3390/cells12081147
Establishment of Skeletal Myogenic Progenitors from Non-Human Primate Induced Pluripotent Stem Cells
Abstract
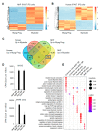
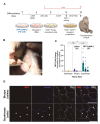
Pluripotent stem (PS) cells enable the scalable production of tissue-specific derivatives with therapeutic potential for various clinical applications, including muscular dystrophies. Given the similarity to human counterparts, the non-human primate (NHP) is an ideal preclinical model to evaluate several questions, including delivery, biodistribution, and immune response. While the generation of human-induced PS (iPS)-cell-derived myogenic progenitors is well established, there have been no data for NHP counterparts, probably due to the lack of an efficient system to differentiate NHP iPS cells towards the skeletal muscle lineage. Here, we report the generation of three independent Macaca fascicularis iPS cell lines and their myogenic differentiation using PAX7 conditional expression. The whole-transcriptome analysis confirmed the successful sequential induction of mesoderm, paraxial mesoderm, and myogenic lineages. NHP myogenic progenitors efficiently gave rise to myotubes under appropriate in vitro differentiation conditions and engrafted in vivo into the TA muscles of NSG and FKRP-NSG mice. Lastly, we explored the preclinical potential of these NHP myogenic progenitors in a single wild-type NHP recipient, demonstrating engraftment and characterizing the interaction with the host immune response. These studies establish an NHP model system through which iPS-cell-derived myogenic progenitors can be studied.
Keywords: RNA sequencing; induced pluripotent stem cells; muscle regeneration; muscular dystrophy; myogenesis; non-human primate; stem cell therapy.
Conflict of interest statement
RCRP is cofounder and holds equity in Myogenica. All other authors declare no conflict of interest.
Figures



References
-
- Azzag K., Ortiz-Cordero C., Oliveira N.A.J., Magli A., Selvaraj S., Tungtur S., Upchurch W., Iaizzo P.A., Lu Q.L., Perlingeiro R.C.R. Efficient engraftment of pluripotent stem cell-derived myogenic progenitors in a novel immunodeficient mouse model of limb girdle muscular dystrophy 2I. Skelet. Muscle. 2020;10:10. doi: 10.1186/s13395-020-00228-3. - DOI - PMC - PubMed
-
- Darabi R., Arpke R.W., Irion S., Dimos J.T., Grskovic M., Kyba M., Perlingeiro R.C. Human ES- and iPS-derived myogenic progenitors restore DYSTROPHIN and improve contractility upon transplantation in dystrophic mice. Cell Stem Cell. 2012;10:610–619. doi: 10.1016/j.stem.2012.02.015. - DOI - PMC - PubMed
-
- Dhoke N.R., Kim H., Selvaraj S., Azzag K., Zhou H., Oliveira N.A., Tungtur S., Ortiz-Cordero C., Kiley J., Lu Q.L., et al. A universal gene correction approach for FKRP-associated dystroglycanopathies to enable autologous cell therapy. Cell Rep. 2021;36:109360. doi: 10.1016/j.celrep.2021.109360. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

