Tertiary Lymphoid Structures (TLSs) and Stromal Blood Vessels Have Significant and Heterogeneous Impact on Recurrence, Lymphovascular and Perineural Invasion amongst Breast Cancer Molecular Subtypes
- PMID: 37190085
- PMCID: PMC10136904
- DOI: 10.3390/cells12081176
Tertiary Lymphoid Structures (TLSs) and Stromal Blood Vessels Have Significant and Heterogeneous Impact on Recurrence, Lymphovascular and Perineural Invasion amongst Breast Cancer Molecular Subtypes
Abstract
Background: Tertiary lymphoid structures (TLSs) mediate local antitumor immunity, and interest in them significantly increased since cancer immunotherapy was implemented. We examined TLS- tumor stromal blood vessel interplay for each breast cancer (BC) molecular subtype related to recurrence, lymphovascular invasion (LVI), and perineural invasion (PnI).
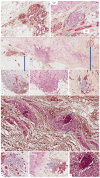
Methods: TLSs were quantified on hematoxylin and eosin stain specimens followed by CD34/smooth muscle actin (SMA) double immunostaining for stromal blood vessel maturation assessment. Statistical analysis linked microscopy to recurrence, LVI, and PnI.
Results: TLS negative (TLS-) subgroups in each BC molecular subtype (except to Luminal A) have higher LVI, PnI, and recurrence. A significant rise in LVI and PnI were observed for the HER2+/TLS- subgroup (p < 0.001). The triple negative breast cancer (TNBC)/TLS- subgroup had the highest recurrence and invasion risk which was also significantly related to tumor grade. PnI but not LVI significantly influenced recurrence in the TNBC/TLS+ subgroup (p < 0.001). TLS-stromal blood vessel interrelation was different amongst BC molecular subtypes.
Conclusion: BC invasion and recurrence are strongly influenced by TLS presence and stromal blood vessels, especially for HER2 and TNBC BC molecular subtypes.
Keywords: TLS; breast cancer molecular subtypes; immature blood vessels; lymphovascular invasion; mature blood vessels; perineural invasion; recurrence; tertiary lymphoid structure.
Conflict of interest statement
The authors have no conflict of interest.
Figures

Similar articles
-
Heterogeneity of the Alpha-Smooth Muscle Actin Tumor Score in Breast Cancer Cells Significantly Affects Tumor Invasiveness, Recurrence, and Patient Survival.Cureus. 2024 Dec 17;16(12):e75908. doi: 10.7759/cureus.75908. eCollection 2024 Dec. Cureus. 2024. PMID: 39698197 Free PMC article.
-
Distinct Tertiary Lymphoid Structure Associations and Their Prognostic Relevance in HER2 Positive and Negative Breast Cancers.Oncologist. 2017 Nov;22(11):1316-1324. doi: 10.1634/theoncologist.2017-0029. Epub 2017 Jul 12. Oncologist. 2017. PMID: 28701569 Free PMC article.
-
Triple-negative breast cancer survival prediction using artificial intelligence through integrated analysis of tertiary lymphoid structures and tumor budding.Cancer. 2024 Apr 15;130(S8):1499-1512. doi: 10.1002/cncr.35261. Epub 2024 Feb 29. Cancer. 2024. PMID: 38422056
-
The Presence of Tertiary Lymphoid Structures Provides New Insight Into the Clinicopathological Features and Prognosis of Patients With Breast Cancer.Front Immunol. 2022 May 19;13:868155. doi: 10.3389/fimmu.2022.868155. eCollection 2022. Front Immunol. 2022. PMID: 35664009 Free PMC article.
-
Tertiary lymphoid structures and cytokines interconnections: The implication in cancer immunotherapy.Cancer Lett. 2023 Aug 1;568:216293. doi: 10.1016/j.canlet.2023.216293. Epub 2023 Jun 29. Cancer Lett. 2023. PMID: 37392991 Review.
Cited by
-
Tertiary lymphoid structures in cancer: immune mechanisms and clinical implications.MedComm (2020). 2024 Mar 11;5(3):e489. doi: 10.1002/mco2.489. eCollection 2024 Mar. MedComm (2020). 2024. PMID: 38469550 Free PMC article. Review.
-
Combined oral low-dose cyclophosphamide endocrine therapy may improve clinical response among patients with metastatic breast cancer via Tregs in TLSs.Sci Rep. 2024 Jun 11;14(1):13432. doi: 10.1038/s41598-024-64042-3. Sci Rep. 2024. PMID: 38862586 Free PMC article.
-
Development of a tertiary lymphoid structure-based prognostic model for breast cancer: integrating single-cell sequencing and machine learning to enhance patient outcomes.Front Immunol. 2025 Feb 26;16:1534928. doi: 10.3389/fimmu.2025.1534928. eCollection 2025. Front Immunol. 2025. PMID: 40078998 Free PMC article.
-
Reassessing Breast Cancer-Associated Fibroblasts (CAFs) Interactions with Other Stromal Components and Clinico-Pathologic Parameters by Using Immunohistochemistry and Digital Image Analysis (DIA).Cancers (Basel). 2023 Jul 27;15(15):3823. doi: 10.3390/cancers15153823. Cancers (Basel). 2023. PMID: 37568639 Free PMC article.
-
Heterogeneity of Cervical Cancer-Associated Tertiary Lymphoid Structures (TLSs) and Their Specific Interrelation With Clinicopathological Parameters.Cureus. 2024 Apr 26;16(4):e59077. doi: 10.7759/cureus.59077. eCollection 2024 Apr. Cureus. 2024. PMID: 38694662 Free PMC article.
References
-
- Dieu-Nosjean M.C., Antoine M., Danel C., Heudes D., Wislez M., Poulot V., Rabbe N., Laurans L., Tartour E., de Chaisemartin L., et al. Long-term survival for patients with non-small-cell lung cancer with intratumoral lymphoid structures. J. Clin. Oncol. 2008;26:4410–4417. doi: 10.1200/JCO.2007.15.0284. - DOI - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

