Current Treatments for Patients with Genetic Obesity
- PMID: 37191347
- PMCID: PMC10234057
- DOI: 10.4274/jcrpe.galenos.2023.2023-3-2
Current Treatments for Patients with Genetic Obesity
Abstract
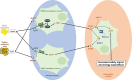
Obesity derives from impaired central control of body weight, implying interaction between environment and an individual genetic predisposition. Genetic obesities, including monogenic and syndromic obesities, are rare and complex neuro-endocrine pathologies where the genetic contribution is predominant. Severe and early-onset obesity with eating disorders associated with frequent comorbidities make these diseases challenging. Their current estimated prevalence of 5-10% in severely obese children is probably underestimated due to the limited access to genetic diagnosis. A central alteration of hypothalamic regulation of weight implies that the leptin-melanocortin pathway is responsible for the symptoms. The management of genetic obesity has so far been only based, above all, on lifestyle intervention, especially regarding nutrition and physical activity. New therapeutic options have emerged in the last years for these patients, raising great hope to manage their complex situation and improve quality of life. Implementation of genetic diagnosis in clinical practice is thus of paramount importance to allow individualized care. This review describes the current clinical management of genetic obesity and the evidence on which it is based. Some insights will also be provided into new therapies under evaluation.
Keywords: Genetic obesity; syndromic obesity; personalized medicine; setmelanotide.
©Copyright 2023 by Turkish Society for Pediatric Endocrinology and Diabetes | The Journal of Clinical Research in Pediatric Endocrinology published by Galenos Publishing House.
Figures

References
-
- Bouchard C. Genetics of Obesity: What We Have Learned Over Decades of Research. Obesity (Silver Spring) 2021;29:802–820. - PubMed
-
- Childhood obesity in European Region remains high: new WHO report presents latest country data [Internet]. 2020 [cited 2022 Dec 23]. Available from: [Internet] https://www.who.int/europe/news/item/08-11-2022-childhood-obesity-in-eur....
-
- WHO. Obesity and overweight [Internet]. 2019 [cited 2022 Dec 21]. Available from: [Internet] https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight.
-
- Silventoinen K, Jelenkovic A, Sund R, Hur YM, Yokoyama Y, Honda C, Hjelmborg Jv, Möller S, Ooki S, Aaltonen S, Ji F, Ning F, Pang Z, Rebato E, Busjahn A, Kandler C, Saudino KJ, Jang KL, Cozen W, Hwang AE, Mack TM, Gao W, Yu C, Li L, Corley RP, Huibregtse BM, Christensen K, Skytthe A, Kyvik KO, Derom CA, Vlietinck RF, Loos RJ, Heikkilä K, Wardle J, Llewellyn CH, Fisher A, McAdams TA, Eley TC, Gregory AM, He M, Ding X, Bjerregaard-Andersen M, Beck-Nielsen H, Sodemann M, Tarnoki AD, Tarnoki DL, Stazi MA, Fagnani C, D’Ippolito C, Knafo-Noam A, Mankuta D, Abramson L, Burt SA, Klump KL, Silberg JL, Eaves LJ, Maes HH, Krueger RF, McGue M, Pahlen S, Gatz M, Butler DA, Bartels M, van Beijsterveldt TC, Craig JM, Saffery R, Freitas DL, Maia JA, Dubois L, Boivin M, Brendgen M, Dionne G, Vitaro F, Martin NG, Medland SE, Montgomery GW, Chong Y, Swan GE, Krasnow R, Magnusson PK, Pedersen NL, Tynelius P, Lichtenstein P, Haworth CM, Plomin R, Bayasgalan G, Narandalai D, Harden KP, Tucker-Drob EM, Öncel SY, Aliev F, Spector T, Mangino M, Lachance G, Baker LA, Tuvblad C, Duncan GE, Buchwald D, Willemsen G, Rasmussen F, Goldberg JH, Sørensen TIa, Boomsma DI, Kaprio J. Genetic and environmental effects on body mass index from infancy to the onset of adulthood: an individual-based pooled analysis of 45 twin cohorts participating in the COllaborative project of Development of Anthropometrical measures in Twins (CODATwins) study. Am J Clin Nutr. 2016;104:371–379. - PMC - PubMed
-
- Wardle J, Carnell S, Haworth CM, Plomin R. Evidence for a strong genetic influence on childhood adiposity despite the force of the obesogenic environment. Am J Clin Nutr. 2008;87:398–404. - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
