The glucagon-like peptide-1 (GLP-1) analogue semaglutide reduces alcohol drinking and modulates central GABA neurotransmission
- PMID: 37192005
- PMCID: PMC10371247
- DOI: 10.1172/jci.insight.170671
The glucagon-like peptide-1 (GLP-1) analogue semaglutide reduces alcohol drinking and modulates central GABA neurotransmission
Abstract
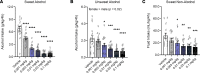
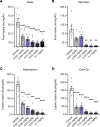
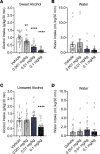
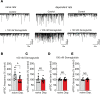
Growing evidence indicates that the glucagon-like peptide-1 (GLP-1) system is involved in the neurobiology of addictive behaviors, and GLP-1 analogues may be used for the treatment of alcohol use disorder (AUD). Here, we examined the effects of semaglutide, a long-acting GLP-1 analogue, on biobehavioral correlates of alcohol use in rodents. A drinking-in-the-dark procedure was used to test the effects of semaglutide on binge-like drinking in male and female mice. We also tested the effects of semaglutide on binge-like and dependence-induced alcohol drinking in male and female rats, as well as acute effects of semaglutide on spontaneous inhibitory postsynaptic currents (sIPSCs) from central amygdala (CeA) and infralimbic cortex (ILC) neurons. Semaglutide dose-dependently reduced binge-like alcohol drinking in mice; a similar effect was observed on the intake of other caloric/noncaloric solutions. Semaglutide also reduced binge-like and dependence-induced alcohol drinking in rats. Semaglutide increased sIPSC frequency in CeA and ILC neurons from alcohol-naive rats, suggesting enhanced GABA release, but had no overall effect on GABA transmission in alcohol-dependent rats. In conclusion, the GLP-1 analogue semaglutide decreased alcohol intake across different drinking models and species and modulated central GABA neurotransmission, providing support for clinical testing of semaglutide as a potentially novel pharmacotherapy for AUD.
Keywords: Addiction; Endocrinology; Neuroscience.
Figures