Bioinspired Active Site with a Coordination-Adaptive Organosulfonate Ligand for Catalytic Water Oxidation at Neutral pH
- PMID: 37196315
- PMCID: PMC10236490
- DOI: 10.1021/jacs.3c03415
Bioinspired Active Site with a Coordination-Adaptive Organosulfonate Ligand for Catalytic Water Oxidation at Neutral pH
Abstract
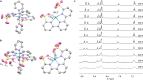
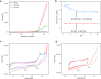
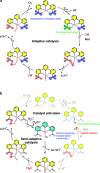
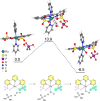
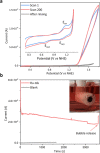
Many enzymes use adaptive frameworks to preorganize substrates, accommodate various structural and electronic demands of intermediates, and accelerate related catalysis. Inspired by biological systems, a Ru-based molecular water oxidation catalyst containing a configurationally labile ligand [2,2':6',2″-terpyridine]-6,6″-disulfonate was designed to mimic enzymatic framework, in which the sulfonate coordination is highly flexible and functions as both an electron donor to stabilize high-valent Ru and a proton acceptor to accelerate water dissociation, thus boosting the catalytic water oxidation performance thermodynamically and kinetically. The combination of single-crystal X-ray analysis, various temperature NMR, electrochemical techniques, and DFT calculations was utilized to investigate the fundamental role of the self-adaptive ligand, demonstrating that the on-demand configurational changes give rise to fast catalytic kinetics with a turnover frequency (TOF) over 2000 s-1, which is compared to oxygen-evolving complex (OEC) in natural photosynthesis.
Conflict of interest statement
The authors declare no competing financial interest.
Figures



References
-
- Ayipo Y. O.; Osunniran W. A.; Babamale H. F.; Ayinde M. O.; Mordi M. N. Metalloenzyme mimicry and modulation strategies to conquer antimicrobial resistance: Metal-ligand coordination perspectives. Coord. Chem. Rev. 2022, 453, 214317 10.1016/j.ccr.2021.214317. - DOI
LinkOut - more resources
Full Text Sources

