CD301b+ macrophage: the new booster for activating bone regeneration in periodontitis treatment
- PMID: 37198150
- PMCID: PMC10192316
- DOI: 10.1038/s41368-023-00225-4
CD301b+ macrophage: the new booster for activating bone regeneration in periodontitis treatment
Abstract
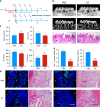
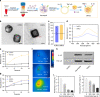
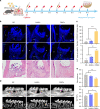
Periodontal bone regeneration is a major challenge in the treatment of periodontitis. Currently the main obstacle is the difficulty of restoring the regenerative vitality of periodontal osteoblast lineages suppressed by inflammation, via conventional treatment. CD301b+ macrophages were recently identified as a subpopulation that is characteristic of a regenerative environment, but their role in periodontal bone repair has not been reported. The current study indicates that CD301b+ macrophages may be a constituent component of periodontal bone repair, and that they are devoted to bone formation in the resolving phase of periodontitis. Transcriptome sequencing suggested that CD301b+ macrophages could positively regulate osteogenesis-related processes. In vitro, CD301b+ macrophages could be induced by interleukin 4 (IL-4) unless proinflammatory cytokines such as interleukin 1β (IL-1β) and tumor necrosis factor α (TNF-α) were present. Mechanistically, CD301b+ macrophages promoted osteoblast differentiation via insulin-like growth factor 1 (IGF-1)/thymoma viral proto-oncogene 1 (Akt)/mammalian target of rapamycin (mTOR) signaling. An osteogenic inducible nano-capsule (OINC) consisting of a gold nanocage loaded with IL-4 as the "core" and mouse neutrophil membrane as the "shell" was designed. When injected into periodontal tissue, OINCs first absorbed proinflammatory cytokines in inflamed periodontal tissue, then released IL-4 controlled by far-red irradiation. These events collectively promoted CD301b+ macrophage enrichment, which further boosted periodontal bone regeneration. The current study highlights the osteoinductive role of CD301b+ macrophages, and suggests a CD301b+ macrophage-targeted induction strategy based on biomimetic nano-capsules for improved therapeutic efficacy, which may also provide a potential therapeutic target and strategy for other inflammatory bone diseases.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Similar articles
-
Tim4 deficiency reduces CD301b+ macrophage and aggravates periodontitis bone loss.Int J Oral Sci. 2024 Feb 28;16(1):20. doi: 10.1038/s41368-023-00270-z. Int J Oral Sci. 2024. PMID: 38418808 Free PMC article.
-
The role of macrophages in the periodontal regeneration using Emdogain gel.J Periodontal Res. 2008 Apr;43(2):143-55. doi: 10.1111/j.1600-0765.2007.01004.x. J Periodontal Res. 2008. PMID: 18302615
-
CD301b+ Macrophages as Potential Target to Improve Orthodontic Treatment under Mild Inflammation.Cells. 2022 Dec 29;12(1):135. doi: 10.3390/cells12010135. Cells. 2022. PMID: 36611929 Free PMC article.
-
Impact of the host response and osteoblast lineage cells on periodontal disease.Front Immunol. 2022 Oct 11;13:998244. doi: 10.3389/fimmu.2022.998244. eCollection 2022. Front Immunol. 2022. PMID: 36304447 Free PMC article. Review.
-
Macrophage Polarization and Osteoporosis: A Review.Nutrients. 2020 Sep 30;12(10):2999. doi: 10.3390/nu12102999. Nutrients. 2020. PMID: 33007863 Free PMC article. Review.
Cited by
-
Advances in hybridized nanoarchitectures for improved oro-dental health.J Nanobiotechnology. 2024 Aug 7;22(1):469. doi: 10.1186/s12951-024-02680-5. J Nanobiotechnology. 2024. PMID: 39113060 Free PMC article. Review.
-
The research progress on periodontitis by the National Natural Science Foundation of China.Int J Oral Sci. 2025 Jun 3;17(1):44. doi: 10.1038/s41368-025-00371-x. Int J Oral Sci. 2025. PMID: 40461458 Free PMC article. Review.
-
Advances in biomaterials for oral-maxillofacial bone regeneration: spotlight on periodontal and alveolar bone strategies.Regen Biomater. 2024 Jul 4;11:rbae078. doi: 10.1093/rb/rbae078. eCollection 2024. Regen Biomater. 2024. PMID: 39055303 Free PMC article. Review.
-
Effects of Insulin-like Growth Factor 1 on the Maintenance of Cell Viability and Osteogenic Differentiation of Gingiva-Derived Mesenchymal Stem Cell Spheroids.Medicina (Kaunas). 2025 Jan 4;61(1):76. doi: 10.3390/medicina61010076. Medicina (Kaunas). 2025. PMID: 39859058 Free PMC article.
-
TRPV1 Regulates Proinflammatory Properties of M1 Macrophages in Periodontitis Via NRF2.Inflammation. 2024 Dec;47(6):2041-2056. doi: 10.1007/s10753-024-02024-3. Epub 2024 May 3. Inflammation. 2024. PMID: 38700791
References
-
- Wu, D. et al. Trem1 induces periodontal inflammation via regulating M1 polarization. J. Dent. Res. 10.1177/00220345211044681 (2021). - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

