Randomized Controlled Trials in ICU in the Four Highest-Impact General Medicine Journals
- PMID: 37199541
- PMCID: PMC10426774
- DOI: 10.1097/CCM.0000000000005937
Randomized Controlled Trials in ICU in the Four Highest-Impact General Medicine Journals
Abstract
Objective: To study ICU trials published in the four highest-impact general medicine journals by comparing them with concurrently published non-ICU trials in the same journals.
Data sources: PubMed was searched for randomized controlled trials (RCTs) published between January 2014 and October 2021 in the New England Journal of Medicine , The Lancet , the Journal of the American Medical Association , and the British Medical Journal.
Study selection: Original RCT publications investigating any type of intervention in any patient population.
Data extraction: ICU RCTs were defined as RCTs exclusively including patients admitted to the ICU. Year and journal of publication, sample size, study design, funding source, study outcome, type of intervention, Fragility Index (FI), and Fragility Quotient were collected.
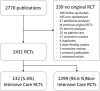
Data synthesis: A total of 2,770 publications were screened. Of 2,431 original RCTs, 132 (5.4%) were ICU RCTs, gradually rising from 4% in 2014 to 7.5% in 2021. ICU RCTs and non-ICU RCTs included a comparable number of patients (634 vs 584, p = 0.528). Notable differences for ICU RCTs were the low occurrence of commercial funding (5% vs 36%, p < 0.001), the low number of RCTs that reached statistical significance (29% vs 65%, p < 0.001), and the low FI when they did reach significance (3 vs 12, p = 0.008).
Conclusions: In the last 8 years, RCTs in ICU medicine made up a meaningful, and growing, portion of RCTs published in high-impact general medicine journals. In comparison with concurrently published RCTs in non-ICU disciplines, statistical significance was rare and often hinged on the outcome events of just a few patients. Increased attention should be paid to realistic expectations of treatment effects when designing ICU RCTs to detect differences in treatment effects that are reliable and clinically relevant.
Copyright © 2023 The Author(s). Published by Wolters Kluwer Health, Inc. on behalf of the Society of Critical Care Medicine and Wolters Kluwer Health, Inc.
Conflict of interest statement
Dr. Hollmann’s institution received funding from International Drug Development Pharma and Medical Development; he received funding as Executive Section Editor of Pharmacology with Anesthesia and Analgesia. The remaining authors have disclosed that they do not have any potential conflicts of interest.
Figures
References
-
- Macleod MR, Michie S, Roberts I, et al. : Biomedical research: Increasing value, reducing waste. Lancet. 2014; 383:101–104 - PubMed
-
- Demarquette A, Perrault T, Alapetite T, et al. : Spin and fragility in randomised controlled trials in the anaesthesia literature: A systematic review. Br J Anaesth. 2023; 130:528–535 - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources