Discovery of 5'-Substituted 5-Fluoro-2'-deoxyuridine Monophosphate Analogs: A Novel Class of Thymidylate Synthase Inhibitors
- PMID: 37200809
- PMCID: PMC10186355
- DOI: 10.1021/acsptsci.2c00252
Discovery of 5'-Substituted 5-Fluoro-2'-deoxyuridine Monophosphate Analogs: A Novel Class of Thymidylate Synthase Inhibitors
Abstract
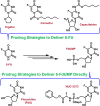
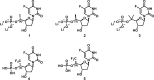
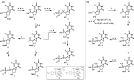
5-Fluorouracil and 5-fluorouracil-based prodrugs have been used clinically for decades to treat cancer. Their anticancer effects are most prominently ascribed to inhibition of thymidylate synthase (TS) by metabolite 5-fluoro-2'-deoxyuridine 5'-monophosphate (FdUMP). However, 5-fluorouracil and FdUMP are subject to numerous unfavorable metabolic events that can drive undesired systemic toxicity. Our previous research on antiviral nucleotides suggested that substitution at the nucleoside 5'-carbon imposes conformational restrictions on the corresponding nucleoside monophosphates, rendering them poor substrates for productive intracellular conversion to viral polymerase-inhibiting triphosphate metabolites. Accordingly, we hypothesized that 5'-substituted analogs of FdUMP, which is uniquely active at the monophosphate stage, would inhibit TS while preventing undesirable metabolism. Free energy perturbation-derived relative binding energy calculations suggested that 5'(R)-CH3 and 5'(S)-CF3 FdUMP analogs would maintain TS potency. Herein, we report our computational design strategy, synthesis of 5'-substituted FdUMP analogs, and pharmacological assessment of TS inhibitory activity.
© 2023 American Chemical Society.
Conflict of interest statement
The authors declare no competing financial interest.
Figures




References
Grants and funding
LinkOut - more resources
Full Text Sources
