COVID-19 Booster Vaccination in Early Pregnancy and Surveillance for Spontaneous Abortion
- PMID: 37204791
- PMCID: PMC10199343
- DOI: 10.1001/jamanetworkopen.2023.14350
COVID-19 Booster Vaccination in Early Pregnancy and Surveillance for Spontaneous Abortion
Abstract
Importance: Adherence to COVID-19 booster vaccine recommendations has lagged in pregnant and nonpregnant adult populations. One barrier to booster vaccination is uncertainty regarding the safety of booster doses among pregnant people.
Objective: To evaluate whether there is an association between COVID-19 booster vaccination during pregnancy and spontaneous abortion.
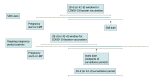
Design, setting, and participants: This observational, case-control, surveillance study evaluated people aged 16 to 49 years with pregnancies at 6 to 19 weeks' gestation at 8 health systems in the Vaccine Safety Datalink from November 1, 2021, to June 12, 2022. Spontaneous abortion cases and ongoing pregnancy controls were evaluated during consecutive surveillance periods, defined by calendar time.
Exposure: Primary exposure was receipt of a third messenger RNA (mRNA) COVID-19 vaccine dose within 28 days before spontaneous abortion or index date (midpoint of surveillance period in ongoing pregnancy controls). Secondary exposures were third mRNA vaccine doses in a 42-day window or any COVID-19 booster in 28- and 42-day windows.
Main outcomes and measures: Spontaneous abortion cases and ongoing pregnancy controls were identified from electronic health data using a validated algorithm. Cases were assigned to a single surveillance period based on pregnancy outcome date. Eligible ongoing pregnancy time was assigned to 1 or more surveillance periods as an ongoing pregnancy-period control. Generalized estimating equations were used to estimate adjusted odds ratios (AOR) with gestational age, maternal age, antenatal visits, race and ethnicity, site, and surveillance period as covariates and robust variance estimates to account for inclusion of multiple pregnancy periods per unique pregnancy.
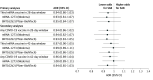
Results: Among 112 718 unique pregnancies included in the study, the mean (SD) maternal age was 30.6 (5.5) years. Pregnant individuals were Asian, non-Hispanic (15.1%); Black, non-Hispanic (7.5%); Hispanic (35.6%); White, non-Hispanic (31.2%); and of other or unknown (10.6%); and 100% were female. Across eight 28-day surveillance periods, among 270 853 ongoing pregnancy-period controls, 11 095 (4.1%) had received a third mRNA COVID-19 vaccine in a 28-day window; among 14 226 cases, 553 (3.9%) had received a third mRNA COVID-19 vaccine within 28 days of the spontaneous abortion. Receipt of a third mRNA COVID-19 vaccine was not associated with spontaneous abortion in a 28-day window (AOR, 0.94; 95% CI, 0.86-1.03). Results were consistent when using a 42-day window (AOR, 0.97; 95% CI, 0.90-1.05) and for any COVID-19 booster in a 28-day (AOR, 0.94; 95% CI, 0.86-1.02) or 42-day (AOR, 0.96; 95% CI, 0.89-1.04) exposure window.
Conclusions and relevance: In this case-control surveillance study, COVID-19 booster vaccination in pregnancy was not associated with spontaneous abortion. These findings support the safety of recommendations for COVID-19 booster vaccination, including in pregnant populations.
Conflict of interest statement
Figures
References
-
- Worldometer COVID-19 coronavirus pandemic . Accessed March 18, 2022. https://worldometers.info/coronavirus/
-
- Strid P, Zapata LB, Tong VT, et al. . Coronavirus disease 2019 (COVID-19) severity among women of reproductive age with symptomatic laboratory—confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection by pregnancy status-United States, 1 January 2020–25 December 2021. Clin Infect Dis. 2022;75(suppl 2):S317-S325. doi:10.1093/cid/ciac479 - DOI - PMC - PubMed
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

