Type I and III interferons shape the retinal cytokine network and barrier function in an in vitro model of ocular toxoplasmosis
- PMID: 37205102
- PMCID: PMC10188120
- DOI: 10.3389/fimmu.2023.1148037
Type I and III interferons shape the retinal cytokine network and barrier function in an in vitro model of ocular toxoplasmosis
Abstract
Introduction: The particularities of the ocular immune environment and its barrier protection in the context of infection are not well elucidated. The apicomplexan parasite Toxoplasma gondii is one of the pathogens successfully crossing this barrier and establishing chronic infection in retinal cells.
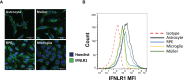
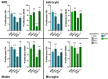
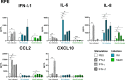
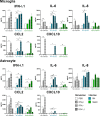
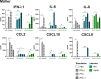
Methods: As a first approach, we studied the initial cytokine network in vitro in four human cell lines: Retinal pigmented epithelial (RPE), microglial, astrocytic and Müller cells. Furthermore, we looked at the consequences of retinal infection on the integrity of the outer blood-retina barrier (oBRB). We particularly focused on the roles of type I and type III interferons, (IFN-β and IFN-λ). Especially IFN-λ is known for its significant role in barrier defense. However, its effect on the retinal barrier or T. gondii infection remains unexplored, unlike IFN-γ, which has been extensively studied in this context.
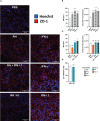
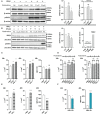
Results and discussion: Here, we show that stimulation with type I and III interferons did not limit parasite proliferation in retinal cells we tested. However, IFN-β and IFN-γ strongly induced inflammatory or cell-attracting cytokine production, whereas IFN-λ1 showed less inflammatory activity. Concomitant T. gondii infection influenced these cytokine patterns, distinctly depending on the parasite strain. Interestingly, all these cells could be stimulated to produce IFN-λ1. Using an in vitro oBRB model based on RPE cells, we observed that interferon stimulation strengthened membrane localization of the tight junction protein ZO-1 and enhanced their barrier function, in a STAT1-independent manner.
Conclusion: Together, our model shows how T. gondii infection shapes the retinal cytokine network and barrier function, and demonstrates the role of type I and type III interferons in these processes.
Keywords: Toxoplasma gondii; barrier function; inflammation; interferons; ocular immunology; retina; tight junctions.
Copyright © 2023 Geiller, Greigert, Hillenbrand, Gommenginger, Beal, Brunet, Filisetti, Villard, Denis and Pfaff.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Weidner JM, Kanatani S, Hernández-Castañeda MA, Fuks JM, Rethi B, Wallin RPA, et al. Rapid cytoskeleton remodelling in dendritic cells following invasion by toxoplasma gondii coincides with the onset of a hypermigratory phenotype. Cell Microbiol (2013) 15(10):1735−52. doi: 10.1111/cmi.12145 - DOI - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

