PD-1 limits differentiation and plasticity of Tc17 cells
- PMID: 37205114
- PMCID: PMC10186197
- DOI: 10.3389/fimmu.2023.1104730
PD-1 limits differentiation and plasticity of Tc17 cells
Abstract
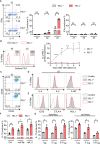
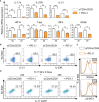
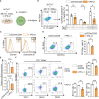
Blockade of surface co-inhibitory receptor programmed cell death-1 (PD-1; CD279) has been established as an important immunotherapeutic approach to treat malignancies. On a cellular level, PD-1 is demonstrated to be of particular importance in inhibiting differentiation and effector function of cytotoxic Tc1 cells (CTLs). Nevertheless, the role of PD-1 in modulating interleukin (IL)-17-producing CD8+ T-cells (Tc17 cells), which generally display suppressed cytotoxic nature, is not well understood. To evaluate the impact of PD-1 in Tc17 responses, we examined its functioning using different in vitro and in vivo models. Upon activation of CD8+ T-cells in Tc17 environment, we found that PD-1 was rapidly expressed on the surface of CD8+ T-cells and triggered a T-cell-internal mechanism that inhibited the expression of IL-17 and Tc17-supporting transcription factors pSTAT3 and RORγt. Expression of type17-polarising cytokine IL-21 and the receptor for IL-23 were also suppressed. Intriguingly, adoptively transferred, PD-1-/- Tc17 cells were highly efficient in rejection of established B16 melanoma in vivo and displayed Tc1 like characteristics ex vivo. When using IL-17A-eGFP reporter mice for in vitro fate tracking, IL-17A-eGFP expressing cells lacking PD-1 signaling upon re-stimulation with IL-12 quickly acquired Tc1 characteristics such as IFN-γ, and granzyme B expression, implicating lineage independent upregulation of CTL-characteristics that are needed for tumor control. In line with plasticity characteristics, absence of PD-1 signaling in Tc17 cells increased the expression of the stemness and persistence-associated molecules TCF1 and BCL6. Thus, PD-1 plays a central role in the specific suppression of Tc17 differentiation and its plasticity in relation to CTL-driven tumor rejection, which provides further explanation as to why the blockade of PD-1 is such an efficient therapeutic target for inducing tumor rejection.
Keywords: T-cell differentiation; T-cell plasticity; Tc17 cells; cytotoxic T lymphocytes (CTLs); immune checkpoint.
Copyright © 2023 Arra, Lingel, Pierau and Brunner-Weinzierl.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
A Th17-like developmental process leads to CD8(+) Tc17 cells with reduced cytotoxic activity.Eur J Immunol. 2009 Jul;39(7):1716-25. doi: 10.1002/eji.200939412. Eur J Immunol. 2009. PMID: 19544308
-
IL-17/IFN-γ double producing CD8+ T (Tc17/IFN-γ) cells: a novel cytotoxic T-cell subset converted from Tc17 cells by IL-12.Int Immunol. 2011 Dec;23(12):751-9. doi: 10.1093/intimm/dxr086. Epub 2011 Oct 29. Int Immunol. 2011. PMID: 22039016
-
CTLA-4 (CD152) enhances the Tc17 differentiation program.Eur J Immunol. 2014 Jul;44(7):2139-52. doi: 10.1002/eji.201343497. Epub 2014 May 30. Eur J Immunol. 2014. PMID: 24723371
-
Tc17 Cells in Immunity and Systemic Autoimmunity.Int Rev Immunol. 2015;34(4):318-31. doi: 10.3109/08830185.2014.954698. Epub 2014 Sep 26. Int Rev Immunol. 2015. PMID: 25259411 Review.
-
IL-17A-producing CD8(+)T cells as therapeutic targets in autoimmunity.Expert Opin Ther Targets. 2015 May;19(5):651-61. doi: 10.1517/14728222.2014.997710. Epub 2015 Jan 22. Expert Opin Ther Targets. 2015. PMID: 25611933 Review.
Cited by
-
PD-1 blockade treatment in melanoma: Mechanism of response and tumor-intrinsic resistance.Cancer Sci. 2025 Feb;116(2):329-337. doi: 10.1111/cas.16398. Epub 2024 Nov 27. Cancer Sci. 2025. PMID: 39601129 Free PMC article. Review.
-
Dual blockade of IL-10 and PD-1 leads to control of SIV viral rebound following analytical treatment interruption.Nat Immunol. 2024 Oct;25(10):1900-1912. doi: 10.1038/s41590-024-01952-4. Epub 2024 Sep 12. Nat Immunol. 2024. PMID: 39266691 Free PMC article.
-
Mouse CD8+ T cell subsets differentially generate IL-17-expressing cells in the colon epithelium and lamina propria.Clin Exp Immunol. 2025 Jan 21;219(1):uxae120. doi: 10.1093/cei/uxae120. Clin Exp Immunol. 2025. PMID: 39745881
-
PD-1 blockade employed at the time CD8+ T cells are activated enhances their antitumor efficacy.J Immunother Cancer. 2025 May 7;13(5):e011145. doi: 10.1136/jitc-2024-011145. J Immunother Cancer. 2025. PMID: 40341032 Free PMC article.
-
CD8 T-cell subsets: heterogeneity, functions, and therapeutic potential.Exp Mol Med. 2023 Nov;55(11):2287-2299. doi: 10.1038/s12276-023-01105-x. Epub 2023 Nov 1. Exp Mol Med. 2023. PMID: 37907738 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

