This is a preprint.
Interaction Between HCN and Slack Channels Regulates mPFC Pyramidal Cell Excitability and Working Memory
- PMID: 37205397
- PMCID: PMC10187370
- DOI: 10.21203/rs.3.rs-2870277/v1
Interaction Between HCN and Slack Channels Regulates mPFC Pyramidal Cell Excitability and Working Memory
Update in
-
Interaction Between HCN and Slack Channels Regulates mPFC Pyramidal Cell Excitability in Working Memory Circuits.Mol Neurobiol. 2024 Apr;61(4):2430-2445. doi: 10.1007/s12035-023-03719-8. Epub 2023 Oct 27. Mol Neurobiol. 2024. PMID: 37889366
Abstract
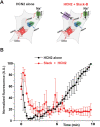
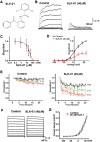
The ability of monkeys and rats to carry out spatial working memory tasks has been shown to depend on the persistent firing of pyramidal cells in the prefrontal cortex (PFC), arising from recurrent excitatory connections on dendritic spines. These spines express hyperpolarization-activated cyclic nucleotide-gated (HCN) channels whose open state is increased by cAMP signaling, and which markedly alter PFC network connectivity and neuronal firing. In traditional neural circuits, activation of these non-selective cation channels leads to neuronal depolarization and increased firing rate. Paradoxically, cAMP activation of HCN channels in PFC pyramidal cells reduces working memory-related neuronal firing. This suggests that activation of HCN channels may hyperpolarize rather than depolarize these neurons. The current study tested the hypothesis that Na+ influx through HCN channels activates Slack Na+-activated K+ (KNa) channels to hyperpolarize the membrane. We have found that HCN and Slack KNa channels coimmunoprecipitate in cortical extracts and that, by immunoelectron microscopy, they colocalize at postsynaptic spines of PFC pyramidal neurons. A specific blocker of HCN channels, ZD7288, reduces KNa current in pyramidal cells that express both HCN and Slack channels, but has no effect on KNa currents in an HEK cell line expressing Slack without HCN channels, indicating that blockade of HCN channels in neurons reduces K+ +current indirectly by lowering Na+ influx. Activation of HCN channels by cAMP in a cell line expressing a Ca2+ reporter results in elevation of cytoplasmic Ca2+, but the effect of cAMP is reversed if the HCN channels are co-expressed with Slack channels. Finally, we used a novel pharmacological blocker of Slack channels to show that inhibition of Slack in rat PFC improves working memory performance, an effect previously demonstrated for blockers of HCN channels. Our results suggest that the regulation of working memory by HCN channels in PFC pyramidal neurons is mediated by an HCN-Slack channel complex that links activation HCN channels to suppression of neuronal excitability.
Keywords: HCN channel; Slack channel; neuronal excitability; working memory.
Conflict of interest statement
Declaration of Interests The authors declare no competing financial interests.
Figures




References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

