Real-world practice of conversion surgery for unresectable hepatocellular carcinoma - a single center data of 26 consecutive patients
- PMID: 37210519
- PMCID: PMC10199571
- DOI: 10.1186/s12885-023-10955-7
Real-world practice of conversion surgery for unresectable hepatocellular carcinoma - a single center data of 26 consecutive patients
Abstract
Aim: To understand the proportion of uHCC (unresectable hepatocellular carcinoma) patients who achieve successful conversion resection in a high-volume setting with state of the art treatment options.
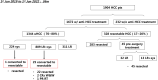
Methods: We retrospectively reviewed all HCC patients hospitalized to our center from June 1st, 2019 to June 1st, 2022. Conversion rate, clinicopathological features, response to systemic and/or loco-regional therapy and surgical outcomes were analyzed.
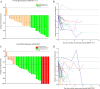
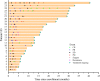
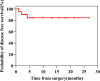
Results: A total of 1,904 HCC patients were identified, with 1672 patients receiving anti-HCC treatment. 328 patients were considered up-front resectable. Of the remaining 1344 uHCC patients, 311 received loco-regional treatment, 224 received systemic treatment, and the remainder (809) received combination systemic plus loco-regional treatment. Following treatment, one patient from the systemic group and 25 patients from the combination group were considered to have resectable disease. A high objective response rate (ORR) was observed in these converted patients (42.3% under RECIST v1.1 and 76.9% under mRECIST criteria). The disease control rate (DCR) reached 100%. 23 patients underwent curative hepatectomy. Major post-operative morbidity was equivalent in the both groups (P=0.76). Pathologic complete response (pCR) was 39.1%. During conversion treatment, grade 3 or higher treatment-related adverse events (TRAEs) were observed in 50% of patients. The median follow-up time was 12.9 months (range, 3.9~40.6) from index diagnosis and 11.4 months (range, 0.9~26.9) from resection. Three patients experienced disease recurrence following conversion surgery.
Conclusions: By intensive treatment, a small sub-group of uHCC patients (2%) may potentially be converted to curative resection. Loco-regional combined with systemic modality was relative safe and effective in the conversion therapy. Short-term outcomes are encouraging, but long-term follow-up in a larger patient population are required to fully understand the utility of this approach.
Keywords: Conversion therapy; Hepatocellular carcinoma; Treatment-related adverse events (TRAEs); Tumor response.
© 2023. The Author(s).
Conflict of interest statement
LZ is on the speakers’ bureau for BeiGene, Bayer, MSD, Roche, Innovent, and Hengrui Medicine. Other authors declare no COI.
Figures
References
-
- Cheng AL, Qin S, Ikeda M, Galle PR, Ducreux M, Kim TY, Lim HY, Kudo M, Breder V, Merle P, et al. Updated efficacy and safety data from IMbrave150: Atezolizumab plus bevacizumab vs. sorafenib for unresectable hepatocellular carcinoma. J Hepatol. 2022;76(4):862–873. doi: 10.1016/j.jhep.2021.11.030. - DOI - PubMed
-
- Finn R, Kudo M, Merle P, Meyer T, Qin S, Ikeda M, Xu R, Edeline J, Ryoo B, Ren Z. LBA34 Primary results from the phase III LEAP-002 study: Lenvatinib plus pembrolizumab versus lenvatinib as first-line (1L) therapy for advanced hepatocellular carcinoma (aHCC) Annals of Oncology. 2022;33:S1401. doi: 10.1016/j.annonc.2022.08.031. - DOI
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

