Establishment and characterization of a new human ampullary carcinoma cell line, DPC-X1
- PMID: 37213400
- PMCID: PMC10198051
- DOI: 10.3748/wjg.v29.i17.2642
Establishment and characterization of a new human ampullary carcinoma cell line, DPC-X1
Abstract
Background: An in-depth study of the pathogenesis and biological characteristics of ampullary carcinoma is necessary to identify appropriate treatment strategies. To date, only eight ampullary cancer cell lines have been reported, and a mixed-type ampullary carcinoma cell line has not yet been reported.
Aim: To establish a stable mixed-type ampullary carcinoma cell line originating from Chinese.
Methods: Fresh ampullary cancer tissue samples were used for primary culture and subculture. The cell line was evaluated by cell proliferation assays, clonal formation assays, karyotype analysis, short tandem repeat (STR) analysis and transmission electron microscopy. Drug resistances against oxaliplatin, paclitaxel, gemcitabine and 5-FU were evaluated by cell counting kit-8 assay. Subcutaneous injection 1 × 106 cells to three BALB/c nude mice for xenograft studies. The hematoxylin-eosin staining was used to detect the pathological status of the cell line. The expression of biomarkers cytokeratin 7 (CK7), cytokeratin 20 (CK20), cytokeratin low molecular weight (CKL), Ki67 and carcinoembryonic antigen (CEA) were determined by immunocytochemistry assay.
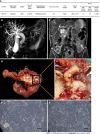
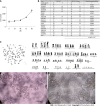
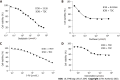
Results: DPC-X1 was continuously cultivated for over a year and stably passaged for more than 80 generations; its population doubling time was 48 h. STR analysis demonstrated that the characteristics of DPC-X1 were highly consistent with those of the patient's primary tumor. Furthermore, karyotype analysis revealed its abnormal sub-tetraploid karyotype. DPC-X1 could efficiently form organoids in suspension culture. Under the transmission electron microscope, microvilli and pseudopods were observed on the cell surface, and desmosomes were visible between the cells. DPC-X1 cells inoculated into BALB/C nude mice quickly formed transplanted tumors, with a tumor formation rate of 100%. Their pathological characteristics were similar to those of the primary tumor. Moreover, DPC-X1 was sensitive to oxaliplatin and paclitaxel and resistant to gemcitabine and 5-FU. Immunohistochemistry showed that the DPC-X1 cells were strongly positive for CK7, CK20, and CKL; the Ki67 was 50%, and CEA was focally expressed.
Conclusion: Here, we have constructed a mixed-type ampullary carcinoma cell line that can be used as an effective model for studying the pathogenesis of ampullary carcinoma and drug development.
Keywords: Ampullary carcinoma; Cell line; Drug resistance; Xenograft.
©The Author(s) 2023. Published by Baishideng Publishing Group Inc. All rights reserved.
Conflict of interest statement
Conflict-of-interest statement: All the authors report no relevant conflicts of interest for this article.
Figures



References
-
- Zheng-Pywell R, Reddy S. Ampullary Cancer. Surg Clin North Am. 2019;99:357–367. - PubMed
-
- Fischer HP, Zhou H. Pathogenesis of carcinoma of the papilla of Vater. J Hepatobiliary Pancreat Surg. 2004;11:301–309. - PubMed
-
- de Castro SM, van Heek NT, Kuhlmann KF, Busch OR, Offerhaus GJ, van Gulik TM, Obertop H, Gouma DJ. Surgical management of neoplasms of the ampulla of Vater: local resection or pancreatoduodenectomy and prognostic factors for survival. Surgery. 2004;136:994–1002. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

