Pooling sputum testing to diagnose tuberculosis using xpert MTB/RIF and xpert ultra: a cost-effectiveness analysis
- PMID: 37217868
- PMCID: PMC10201466
- DOI: 10.1186/s12879-023-08330-9
Pooling sputum testing to diagnose tuberculosis using xpert MTB/RIF and xpert ultra: a cost-effectiveness analysis
Abstract
Background: The World Health Organization (WHO) recommends the diagnosis of tuberculosis (TB) using molecular tests, such as Xpert MTB/RIF (MTB/RIF) or Xpert Ultra (Ultra). These tests are expensive and resource-consuming, and cost-effective approaches are needed for greater coverage.
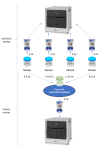
Methods: We evaluated the cost-effectiveness of pooling sputum samples for TB testing by using a fixed amount of 1,000 MTB/RIF or Ultra cartridges. We used the number of people with TB detected as the indicator for cost-effectiveness. Cost-minimization analysis was conducted from the healthcare system perspective and included the costs to the healthcare system using pooled and individual testing.
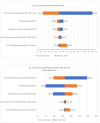
Results: There was no significant difference in the overall performance of the pooled testing using MTB/RIF or Ultra (sensitivity, 93.9% vs. 97.6%, specificity 98% vs. 97%, p-value > 0.1 for both). The mean unit cost across all studies to test one person was 34.10 international dollars for the individual testing and 21.95 international dollars for the pooled testing, resulting in a savings of 12.15 international dollars per test performed (35.6% decrease). The mean unit cost per bacteriologically confirmed TB case was 249.64 international dollars for the individual testing and 162.44 international dollars for the pooled testing (34.9% decrease). Cost-minimization analysis indicates savings are directly associated with the proportion of samples that are positive. If the TB prevalence is ≥ 30%, pooled testing is not cost-effective.
Conclusion: Pooled sputum testing can be a cost-effective strategy for diagnosis of TB, resulting in significant resource savings. This approach could increase testing capacity and affordability in resource-limited settings and support increased testing towards achievement of WHO End TB strategy.
Keywords: Cost-effectiveness; Cost-minimization analysis; Pooling; Tuberculosis; Xpert MTB/RIF; Xpert Ultra.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Pooled testing of sputum with Xpert MTB/RIF and Xpert Ultra during tuberculosis active case finding campaigns in Lao People's Democratic Republic.BMJ Glob Health. 2022 Feb;7(2):e007592. doi: 10.1136/bmjgh-2021-007592. BMJ Glob Health. 2022. PMID: 35165095 Free PMC article.
-
The New Xpert MTB/RIF Ultra: Improving Detection of Mycobacterium tuberculosis and Resistance to Rifampin in an Assay Suitable for Point-of-Care Testing.mBio. 2017 Aug 29;8(4):e00812-17. doi: 10.1128/mBio.00812-17. mBio. 2017. PMID: 28851844 Free PMC article.
-
Xpert MTB/RIF and Xpert MTB/RIF Ultra assays for active tuberculosis and rifampicin resistance in children.Cochrane Database Syst Rev. 2020 Aug 27;8(8):CD013359. doi: 10.1002/14651858.CD013359.pub2. Cochrane Database Syst Rev. 2020. Update in: Cochrane Database Syst Rev. 2022 Sep 6;9:CD013359. doi: 10.1002/14651858.CD013359.pub3. PMID: 32853411 Free PMC article. Updated.
-
Xpert MTB/RIF and Xpert Ultra assays for screening for pulmonary tuberculosis and rifampicin resistance in adults, irrespective of signs or symptoms.Cochrane Database Syst Rev. 2021 Mar 23;3(3):CD013694. doi: 10.1002/14651858.CD013694.pub2. Cochrane Database Syst Rev. 2021. PMID: 33755189 Free PMC article.
-
Xpert MTB/RIF Ultra and Xpert MTB/RIF assays for extrapulmonary tuberculosis and rifampicin resistance in adults.Cochrane Database Syst Rev. 2021 Jan 15;1(1):CD012768. doi: 10.1002/14651858.CD012768.pub3. Cochrane Database Syst Rev. 2021. PMID: 33448348 Free PMC article.
Cited by
-
Diagnostic performance of Ziehl-Neelsen staining and Auramine-Rhodamine staining techniques in the detection of pulmonary and extrapulmonary tuberculosis.Rev Peru Med Exp Salud Publica. 2025 Jun 9;42(1):37-45. doi: 10.17843/rpmesp.2025.421.14062. Rev Peru Med Exp Salud Publica. 2025. PMID: 40498931 Free PMC article.
-
Expanding molecular diagnostic coverage for tuberculosis by combining computer-aided chest radiography and sputum specimen pooling: a modeling study from four high-burden countries.BMC Glob Public Health. 2024;2(1):52. doi: 10.1186/s44263-024-00081-2. Epub 2024 Aug 1. BMC Glob Public Health. 2024. PMID: 39100507 Free PMC article.
-
Implementation of large-scale pooled testing to increase rapid molecular diagnostic test coverage for tuberculosis: a retrospective evaluation.Sci Rep. 2023 Sep 16;13(1):15358. doi: 10.1038/s41598-023-41904-w. Sci Rep. 2023. PMID: 37717043 Free PMC article.
-
The Role of GeneXpert® for Tuberculosis Diagnostics in Brazil: An Examination from a Historical and Epidemiological Perspective.Trop Med Infect Dis. 2023 Oct 26;8(11):483. doi: 10.3390/tropicalmed8110483. Trop Med Infect Dis. 2023. PMID: 37999602 Free PMC article.
References
-
- World Health Organization. Global tuberculosis report 2021. 2021.
-
- World Health Organization. WHO consolidated guidelines on tuberculosis. Module 3: diagnosis - rapid diagnostics for tuberculosis detection, 2021 update. 2021.
-
- World Health Organization. WHO meeting report of a technical expert consultation: non-inferiority analysis of Xpert MTF/RIF Ultra compared to Xpert MTB/RIF. 2017.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical