Adolescent stress impairs postpartum social behavior via anterior insula-prelimbic pathway in mice
- PMID: 37221211
- PMCID: PMC10205810
- DOI: 10.1038/s41467-023-38799-6
Adolescent stress impairs postpartum social behavior via anterior insula-prelimbic pathway in mice
Abstract
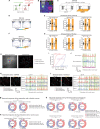
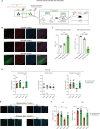
Adolescent stress can be a risk factor for abnormal social behavior in the postpartum period, which critically affects an individual social functioning. Nonetheless, the underlying mechanisms remain unclear. Using a mouse model with optogenetics and in vivo calcium imaging, we found that adolescent psychosocial stress, combined with pregnancy and delivery, caused hypofunction of the glutamatergic pathway from the anterior insula to prelimbic cortex (AI-PrL pathway), which altered PrL neuronal activity, and in turn led to abnormal social behavior. Specifically, the AI-PrL pathway played a crucial role during recognizing the novelty of other mice by modulating "stable neurons" in PrL, which were constantly activated or inhibited by novel mice. We also observed that glucocorticoid receptor signaling in the AI-PrL pathway had a causal role in stress-induced postpartum changes. Our findings provide functional insights into a cortico-cortical pathway underlying adolescent stress-induced postpartum social behavioral deficits.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures



Update of
-
Adolescent stress impairs postpartum social behavior via anterior insula-prelimbic pathway.bioRxiv [Preprint]. 2023 Apr 5:2023.01.03.522598. doi: 10.1101/2023.01.03.522598. bioRxiv. 2023. Update in: Nat Commun. 2023 May 23;14(1):2975. doi: 10.1038/s41467-023-38799-6. PMID: 36711960 Free PMC article. Updated. Preprint.
Similar articles
-
Adolescent stress impairs postpartum social behavior via anterior insula-prelimbic pathway.bioRxiv [Preprint]. 2023 Apr 5:2023.01.03.522598. doi: 10.1101/2023.01.03.522598. bioRxiv. 2023. Update in: Nat Commun. 2023 May 23;14(1):2975. doi: 10.1038/s41467-023-38799-6. PMID: 36711960 Free PMC article. Updated. Preprint.
-
Encoding of social novelty by sparse GABAergic neural ensembles in the prelimbic cortex.Sci Adv. 2022 Sep 2;8(35):eabo4884. doi: 10.1126/sciadv.abo4884. Epub 2022 Aug 31. Sci Adv. 2022. PMID: 36044579 Free PMC article.
-
Divergent Prelimbic Cortical Pathways Interact with BDNF to Regulate Cocaine-seeking.J Neurosci. 2018 Oct 17;38(42):8956-8966. doi: 10.1523/JNEUROSCI.1332-18.2018. Epub 2018 Sep 5. J Neurosci. 2018. PMID: 30185459 Free PMC article.
-
The Role of the Insular Cortex in Pain.Int J Mol Sci. 2023 Mar 17;24(6):5736. doi: 10.3390/ijms24065736. Int J Mol Sci. 2023. PMID: 36982807 Free PMC article. Review.
-
The role of anterior insular cortex in social emotions.Brain Struct Funct. 2010 Jun;214(5-6):579-91. doi: 10.1007/s00429-010-0251-3. Epub 2010 Apr 29. Brain Struct Funct. 2010. PMID: 20428887 Review.
Cited by
-
Prolonged HPA axis dysregulation in postpartum depression associated with adverse early life experiences: A cross-species translational study.Nat Ment Health. 2024 May;2(5):593-604. doi: 10.1038/s44220-024-00217-1. Epub 2024 Apr 11. Nat Ment Health. 2024. PMID: 38736646 Free PMC article.
-
A vagus nerve dominant tetra-synaptic ascending pathway for gastric pain processing.Nat Commun. 2024 Nov 13;15(1):9824. doi: 10.1038/s41467-024-54056-w. Nat Commun. 2024. PMID: 39537596 Free PMC article.
-
Acetylcholine modulates prefrontal outcome coding during threat learning under uncertainty.Elife. 2025 Mar 5;13:RP102986. doi: 10.7554/eLife.102986. Elife. 2025. PMID: 40042523 Free PMC article.
-
Adolescent stress accelerates postpartum novelty recognition impairment in 5xFAD mice.Front Neurosci. 2024 May 15;18:1366199. doi: 10.3389/fnins.2024.1366199. eCollection 2024. Front Neurosci. 2024. PMID: 38812977 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

