Effective dose window for containing tumor burden under tolerable level
- PMID: 37221258
- PMCID: PMC10205748
- DOI: 10.1038/s41540-023-00279-4
Effective dose window for containing tumor burden under tolerable level
Abstract
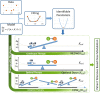
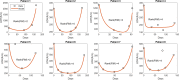
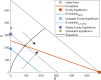
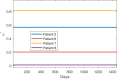
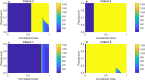
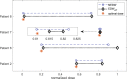
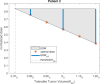
A maximum-tolerated dose (MTD) reduces the drug-sensitive cell population, though it may result in the competitive release of drug resistance. Alternative treatment strategies such as adaptive therapy (AT) or dose modulation aim to impose competitive stress on drug-resistant cell populations by maintaining a sufficient number of drug-sensitive cells. However, given the heterogeneous treatment response and tolerable tumor burden level of individual patients, determining an effective dose that can fine-tune competitive stress remains challenging. This study presents a mathematical model-driven approach that determines the plausible existence of an effective dose window (EDW) as a range of doses that conserve sufficient sensitive cells while maintaining the tumor volume below a threshold tolerable tumor volume (TTV). We use a mathematical model that explains intratumor cell competition. Analyzing the model, we derive an EDW determined by TTV and the competitive strength. By applying a fixed endpoint optimal control model, we determine the minimal dose to contain cancer at a TTV. As a proof of concept, we study the existence of EDW for a small cohort of melanoma patients by fitting the model to longitudinal tumor response data. We performed identifiability analysis, and for the patients with uniquely identifiable parameters, we deduced patient-specific EDW and minimal dose. The tumor volume for a patient could be theoretically contained at the TTV either using continuous dose or AT strategy with doses belonging to EDW. Further, we conclude that the lower bound of the EDW approximates the minimum effective dose (MED) for containing tumor volume at the TTV.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Adaptive Therapy for Metastatic Melanoma: Predictions from Patient Calibrated Mathematical Models.Cancers (Basel). 2021 Feb 16;13(4):823. doi: 10.3390/cancers13040823. Cancers (Basel). 2021. PMID: 33669315 Free PMC article.
-
The Effect of Total Tumor Volume on the Biologically Effective Dose to Tumor and Kidneys for 177Lu-Labeled PSMA Peptides.J Nucl Med. 2018 Jun;59(6):929-933. doi: 10.2967/jnumed.117.203505. Epub 2018 Feb 1. J Nucl Med. 2018. PMID: 29419479
-
Poster - Thur Eve - 60: Physical and dynamic wedges in radiotherapy for rectal cancer: A dosimetric comparison.Med Phys. 2012 Jul;39(7Part3):4636. doi: 10.1118/1.4740168. Med Phys. 2012. PMID: 28516699
-
Comparison of survival rates as predicted by total tumor volume or tumor burden score in patients with hepatocellular carcinoma concurrent with fatty liver disease and hepatitis B virus.Expert Rev Gastroenterol Hepatol. 2023 May;17(5):499-507. doi: 10.1080/17474124.2023.2196403. Epub 2023 Apr 3. Expert Rev Gastroenterol Hepatol. 2023. PMID: 36975382
-
Significance of particle parameters in the evaluation of exposure-dose-response relationships of inhaled particles.Inhal Toxicol. 1996;8 Suppl:73-89. Inhal Toxicol. 1996. PMID: 11542496 Review.
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical
Research Materials

