Additive Engineering for Stable and Efficient Dion-Jacobson Phase Perovskite Solar Cells
- PMID: 37221320
- PMCID: PMC10205963
- DOI: 10.1007/s40820-023-01110-9
Additive Engineering for Stable and Efficient Dion-Jacobson Phase Perovskite Solar Cells
Abstract
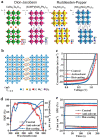
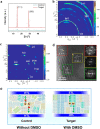
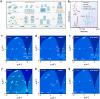
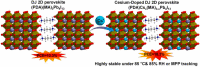
Because of their better chemical stability and fascinating anisotropic characteristics, Dion-Jacobson (DJ)-layered halide perovskites, which owe crystallographic two-dimensional structures, have fascinated growing attention for solar devices. DJ-layered halide perovskites have special structural and photoelectronic features that allow the van der Waals gap to be eliminated or reduced. DJ-layered halide perovskites have improved photophysical characteristics, resulting in improved photovoltaic performance. Nevertheless, owing to the nature of the solution procedure and the fast crystal development of DJ perovskite thin layers, the precursor compositions and processing circumstances can cause a variety of defects to occur. The application of additives can impact DJ perovskite crystallization and film generation, trap passivation in the bulk and/or at the surface, interface structure, and energetic tuning. This study discusses recent developments in additive engineering for DJ multilayer halide perovskite film production. Several additive-assisted bulk and interface optimization methodologies are summarized. Lastly, an overview of research developments in additive engineering in the production of DJ-layered halide perovskite solar cells is offered.
Keywords: Additive compounds; Defect passivation; Dion–Jacobson phases; Perovskite solar cells; Stability.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no conflict of interest.
Figures









References
-
- Dahal B, Li W. Configuration of methylammonium lead iodide perovskite solar cell and its effect on the device's performance: a review. Adv. Mater. Interfaces. 2022;9(19):2200042. doi: 10.1002/admi.202200042. - DOI
-
- Dou Q, Whatley T, Syed T, Wei W, Wang H. Carbon nanomaterials-polymer composites for perovskite solar cells: preparation, properties and applications. J. Mater. Chem. A. 2022;10(37):19211–19230. doi: 10.1039/d2ta02175g. - DOI
-
- E.C. Kohlrausch, D.d.V. Freitas, C.I. da Silva Filho, L.F. Loguercio, L.A. Santa-Cruz et al., Advances in carbon materials applied to carbon-based perovskite solar cells. Energy Technol. 2200676 (2023). 10.1002/ente.202200676
-
- Luo X, Lin X, Gao F, Zhao Y, Li X, et al. Recent progress in perovskite solar cells: from device to commercialization. Sci. China-Chem. 2022;65(12):2369–2416. doi: 10.1007/s11426-022-1426-x. - DOI
-
- Sharma D, Mehra R, Raj B. Comparative study of hole transporting layers commonly used in high-efficiency perovskite solar cells. J. Mater. Sci. 2022;57(45):21172–21191. doi: 10.1007/s10853-022-07958-3. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous
