Clinical Use of On-Demand Therapies for Patients with Parkinson's Disease and OFF Periods
- PMID: 37221354
- PMCID: PMC10310675
- DOI: 10.1007/s40120-023-00486-5
Clinical Use of On-Demand Therapies for Patients with Parkinson's Disease and OFF Periods
Abstract
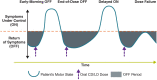
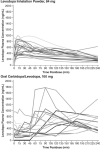
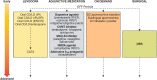
On-demand therapies for Parkinson's disease (PD) provide rapid, reliable relief for patients experiencing OFF periods; however, practical guidelines on the use of these therapies are not generally available. This paper reviews the use of on-demand treatments. Motor fluctuations occur in nearly all patients with PD after long-term use of levodopa. As the goal of PD treatment is to provide good ON time, on-demand treatments that have a more rapid reliable onset than the slower-acting oral medications provide rapid relief for OFF periods. All current on-demand treatments bypass the gastrointestinal tract, providing dopaminergic therapy directly into the blood stream by subcutaneous injection, through the buccal mucosa, or by inhalation into the pulmonary circulation. On-demand treatments are fast acting (10- to 20-min onset), with maximum, reliable, and significant responses reached within 30 min after administration. Oral medications pass through the gastrointestinal tract and thus have slower absorption owing to gastroparesis and competition with food. On-demand therapies, by providing fast-acting relief, can have a positive impact on a patient's quality of life when patients are experiencing OFF periods.
Keywords: Apomorphine; Levodopa; On-demand therapy; Parkinson’s disease; Rescue therapy.
© 2023. The Author(s).
Conflict of interest statement
Rajesh Pahwa has received consulting fees from Abbott, AbbVie, Acadia, Acorda, Adamas, Cala Health, Global Kinetics, Impel Neuropharma, Lundbeck, Neurocrine, Orbis Bioscience, PhotoPharmics, Prilenia, Sunovion, Teva Neuroscience, and US WorldMeds. He also received research support from Abbott, AbbVie, Acorda, Biogen, Boston Scientific, Cala Health, Cavion, Cynapsus, Intec, Kyowa, Lilly, NIH/NINDS, NPF, PSG, Roche, Sunovion, Theranexus, Theravance, US WorldMeds, and Voyager. Fernando L. Pagan is a consultant/speaker for AbbVie, Acadia, Acorda, Adamas, Kyowa Kirin, Lundbeck, Merz, Neurocrine, Sunovion, Supernus, Teva Neuroscience, and US WorldMeds. He is a board member and cofounder of Keiferx and has received educational/research grants from Medtronic and US WorldMeds. Daniel E. Kremens is a consultant and speakers bureau member for Acadia, Adamas, Impax, Lundbeck, Teva, UCB, and US WorldMeds; a consultant for AbbVie, Allergan, GE Healthcare, Kyowa, Merz, Neurocrine, St. Jude Medical, and Sunovion; a researcher for Enterin and Revance; and a researcher and consultant for Acorda. Marie Saint-Hilaire is a consultant for Acorda and Watermark; has received research support from NIH, Michael J. Fox Foundation, American Parkinson Disease Association, Biogen, Neuraly, and Pharma Two B.
Figures

Similar articles
-
On demand therapy for Parkinson's disease patients: Opportunities and choices.Postgrad Med. 2021 Sep;133(7):721-727. doi: 10.1080/00325481.2021.1936087. Epub 2021 Jun 30. Postgrad Med. 2021. PMID: 34082655 Review.
-
Should "on-demand" treatments for Parkinson's disease OFF episodes be used earlier?Clin Park Relat Disord. 2022 Aug 12;7:100161. doi: 10.1016/j.prdoa.2022.100161. eCollection 2022. Clin Park Relat Disord. 2022. PMID: 36033905 Free PMC article. Review.
-
On-Demand Therapy for OFF Episodes in Parkinson's Disease.Mov Disord. 2021 Oct;36(10):2244-2253. doi: 10.1002/mds.28726. Epub 2021 Aug 7. Mov Disord. 2021. PMID: 34363424 Review.
-
Expert Consensus Group report on the use of apomorphine in the treatment of Parkinson's disease--Clinical practice recommendations.Parkinsonism Relat Disord. 2015 Sep;21(9):1023-30. doi: 10.1016/j.parkreldis.2015.06.012. Epub 2015 Jun 17. Parkinsonism Relat Disord. 2015. PMID: 26189414 Review.
-
A technology evaluation of CVT-301 (Inbrija): an inhalable therapy for treatment of Parkinson's disease.Expert Opin Drug Deliv. 2021 Nov;18(11):1559-1569. doi: 10.1080/17425247.2021.1960820. Epub 2021 Sep 10. Expert Opin Drug Deliv. 2021. PMID: 34311641
Cited by
-
Cost of Quality of Life in Advanced Parkinson's Disease: Efficient Strategies for Disease Assessment.Rev Neurol. 2025 Jul 23;80(6):33482. doi: 10.31083/RN33482. Rev Neurol. 2025. PMID: 40767109 Free PMC article. English.
-
Motor and non-motor fluctuations in Parkinson's disease: the knowns and unknowns of current therapeutic approaches.J Neural Transm (Vienna). 2025 Jul 28. doi: 10.1007/s00702-025-02990-4. Online ahead of print. J Neural Transm (Vienna). 2025. PMID: 40719828 Review.
-
Real-world experience with continuous subcutaneous foslevodopa/foscarbidopa infusion: insights and recommendations.J Neural Transm (Vienna). 2025 Mar 22. doi: 10.1007/s00702-025-02911-5. Online ahead of print. J Neural Transm (Vienna). 2025. PMID: 40121314
References
-
- Hauser RA, LeWitt PA, Comella CL. On demand therapy for Parkinson's disease patients: opportunities and choices. Postgrad Med. 2021;133(7):721–727. - PubMed
-
- Olanow CW, Poewe W, Rascol O, Stocchi F. On-demand therapy for OFF episodes in Parkinson's disease. Mov Disord. 2021;36(10):2244–2253. - PubMed
-
- Connolly BS, Lang AE. Pharmacological treatment of Parkinson disease: a review. JAMA. 2014;311(16):1670–1683. - PubMed
-
- Supernus Pharmaceuticals. Apokyn® (apomorphine hydrochloride injection). Full Prescribing Information. Supernus Pharmaceuticals; 2020.
-
- Pahwa R, Factor SA, Lyons KE, et al. Practice parameter: treatment of Parkinson disease with motor fluctuations and dyskinesia (an evidence-based review): report of the Quality Standards Subcommittee of the American Academy of Neurology. Neurology. 2006;66(7):983–995. - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

