Molecular and Neural Mechanisms of Temperature Preference Rhythm in Drosophila melanogaster
- PMID: 37222551
- PMCID: PMC10330063
- DOI: 10.1177/07487304231171624
Molecular and Neural Mechanisms of Temperature Preference Rhythm in Drosophila melanogaster
Abstract
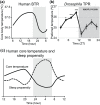
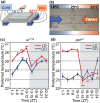
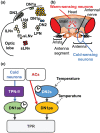
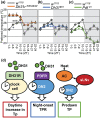
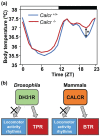
Temperature influences animal physiology and behavior. Animals must set an appropriate body temperature to maintain homeostasis and maximize survival. Mammals set their body temperatures using metabolic and behavioral strategies. The daily fluctuation in body temperature is called the body temperature rhythm (BTR). For example, human body temperature increases during wakefulness and decreases during sleep. BTR is controlled by the circadian clock, is closely linked with metabolism and sleep, and entrains peripheral clocks located in the liver and lungs. However, the underlying mechanisms of BTR are largely unclear. In contrast to mammals, small ectotherms, such as Drosophila, control their body temperatures by choosing appropriate environmental temperatures. The preferred temperature of Drosophila increases during the day and decreases at night; this pattern is referred to as the temperature preference rhythm (TPR). As flies are small ectotherms, their body temperature is close to that of the surrounding environment. Thus, Drosophila TPR produces BTR, which exhibits a pattern similar to that of human BTR. In this review, we summarize the regulatory mechanisms of TPR, including recent studies that describe neuronal circuits relaying ambient temperature information to dorsal neurons (DNs). The neuropeptide diuretic hormone 31 (DH31) and its receptor (DH31R) regulate TPR, and a mammalian homolog of DH31R, the calcitonin receptor (CALCR), also plays an important role in mouse BTR regulation. In addition, both fly TPR and mammalian BTR are separately regulated from another clock output, locomotor activity rhythms. These findings suggest that the fundamental mechanisms of BTR regulation may be conserved between mammals and flies. Furthermore, we discuss the relationships between TPR and other physiological functions, such as sleep. The dissection of the regulatory mechanisms of Drosophila TPR could facilitate an understanding of mammalian BTR and the interaction between BTR and sleep regulation.
Keywords: Drosophila melanogaster; body temperature rhythm; calcitonin receptor; circadian clock; circadian rhythms; diuretic hormone 31; dorsal neurons; temperature preference rhythm.
Conflict of interest statement
The authors have no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Figures
Similar articles
-
Drosophila Temperature Preference Rhythms: An Innovative Model to Understand Body Temperature Rhythms.Int J Mol Sci. 2019 Apr 23;20(8):1988. doi: 10.3390/ijms20081988. Int J Mol Sci. 2019. PMID: 31018551 Free PMC article. Review.
-
Drosophila DH31 Neuropeptide and PDF Receptor Regulate Night-Onset Temperature Preference.J Neurosci. 2016 Nov 16;36(46):11739-11754. doi: 10.1523/JNEUROSCI.0964-16.2016. J Neurosci. 2016. PMID: 27852781 Free PMC article.
-
Calcitonin receptors are ancient modulators for rhythms of preferential temperature in insects and body temperature in mammals.Genes Dev. 2018 Jan 15;32(2):140-155. doi: 10.1101/gad.307884.117. Epub 2018 Feb 12. Genes Dev. 2018. PMID: 29440246 Free PMC article.
-
Circadian rhythm of temperature preference and its neural control in Drosophila.Curr Biol. 2012 Oct 9;22(19):1851-7. doi: 10.1016/j.cub.2012.08.006. Epub 2012 Sep 13. Curr Biol. 2012. PMID: 22981774 Free PMC article.
-
Circadian Rhythms and Sleep in Drosophila melanogaster.Genetics. 2017 Apr;205(4):1373-1397. doi: 10.1534/genetics.115.185157. Genetics. 2017. PMID: 28360128 Free PMC article. Review.
Cited by
-
Thermosensation and Temperature Preference: From Molecules to Neuronal Circuits in Drosophila.Cells. 2023 Dec 8;12(24):2792. doi: 10.3390/cells12242792. Cells. 2023. PMID: 38132112 Free PMC article. Review.
-
Cellular and molecular organization of the Drosophila foregut.Proc Natl Acad Sci U S A. 2024 Mar 12;121(11):e2318760121. doi: 10.1073/pnas.2318760121. Epub 2024 Mar 5. Proc Natl Acad Sci U S A. 2024. PMID: 38442150 Free PMC article.
-
Taste triggers a homeostatic temperature control in hungry flies.Elife. 2024 Dec 2;13:RP94703. doi: 10.7554/eLife.94703. Elife. 2024. PMID: 39621014 Free PMC article.
References
-
- Becskei C, Riediger T, Zund D, Wookey P, Lutz TA. (2004) Immunohistochemical mapping of calcitonin receptors in the adult rat brain. Brain Res 1030:221-233. - PubMed
-
- Ben-Hamo M, Pinshow B, McCue MD, McWilliams SR, Bauchinger U. (2010) Fasting triggers hypothermia, and ambient temperature modulates its depth in Japanese quail Coturnix japonica. Comp Biochem Physiol A Mol Integr Physiol 156:84-91. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Research Materials

