Establishing the Australian National Endometriosis Clinical and Scientific Trials (NECST) Registry: A protocol paper
- PMID: 37224076
- PMCID: PMC10305626
- DOI: 10.1530/RAF-23-0014
Establishing the Australian National Endometriosis Clinical and Scientific Trials (NECST) Registry: A protocol paper
Abstract
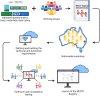
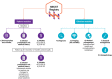
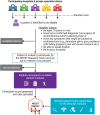
Endometriosis is a common yet under-recognised chronic inflammatory disease, affecting 176 million women, trans and gender diverse people globally. The National Endometriosis Clinical and Scientific Trials (NECST) Registry is a new clinical registry, collecting and tracking diagnostic and treatment data, and patient-reported outcomes on people with endometriosis. The registry is a research priority action item from the 2018 National Action Plan for Endometriosis and aims to provide, large-scale, national and longitudinal population-based data on endometriosis. Working groups (consisting of patients with endometriosis, clinicians and researchers) developing the NECST Registry data dictionary and data collection platform started in 2019. Our data dictionary was developed based on existing and validated questionnaires, tools, meta-data and data cubes - World Endometriosis Research Foundation (WERF) Endometriosis Phenome and Biobanking Harmonisation Project (EPHect), endometriosis CORE outcomes set, patient-reported outcome measures, the International Statistical Classification of Diseases-10th Revision Australian Modification diagnosis codes, and Australian Government datasets: Australian Institute for Health and Welfare (for sociodemographic data), Medicare Benefits Schedule (MBS; for medical procedures) and the Pharmaceutical Benefits Scheme (PBS; for medical therapies). The resulting NECST Registry is an online, secure cloud-based database; prospectively collecting minimum core clinical and health data across eight patient and clinician modules and longitudinal data tracking disease life course. The NECST Registry has ethics approval (HREC/62508/MonH-2020) and is registered on the Australian New Zealand Clinical Trials Registry (ACTRN12622000987763).
Conflict of interest statement
CN reports grant funding from Medical Research Future Fund (MRFF) and other financial or non-financial interests with CSL Vifor as a previous employee (formerly Vifor Pharma Pty. Ltd.; past employee). GDM reports grant funding from NHMRC. PR reports grant funding from MRFF; Participation on a Data Safety Monitoring Board or Advisory Board with Bayer; Leadership or fiduciary role in other board, society, committee or advocacy group with Jean Hailes for Women’s Health (Board Member); and other financial or non-financial interests with University of Melbourne and Royal Women’s Hospital (employee). JA reports grant funding from MRFF; honoraria for lectures, presentations, speakers bureaus, manuscript writing or educational events from Hologic Australia; Participation on a Data Safety Monitoring Board or Advisory Board with Hologic Australia and CSL Vifor (formerly Vifor Pharma Pty. Ltd.); Leadership roles with the Australasian Gynaecological Endoscopy & Surgery Society (AGES; Past president), Heavy Menstrual Bleeding Clinical Care Standards 2017 and 2023 (Australian Government; Co-Chair), Expert Endometriosis Working Group and Endometriosis Clinical Guidelines Group (RANZCOG, Chair); Endometriosis Australia (Former Chair, 2021–2022 (Associate Editor). The other authors have no conflicts of interest to declare.
Figures
Similar articles
-
Protocol for a case-control study investigating the clinical phenotypes and genetic regulation of endometriosis in Indian women: the ECGRI study.BMJ Open. 2021 Aug 9;11(8):e050844. doi: 10.1136/bmjopen-2021-050844. BMJ Open. 2021. PMID: 34373312 Free PMC article.
-
Protocol for the Cultural Translation and Adaptation of the World Endometriosis Research Foundation Endometriosis Phenome and Biobanking Harmonization Project Endometriosis Participant Questionnaire (EPHect).Front Glob Womens Health. 2021 Mar 26;2:644609. doi: 10.3389/fgwh.2021.644609. eCollection 2021. Front Glob Womens Health. 2021. PMID: 34816197 Free PMC article.
-
World Endometriosis Society consensus on the classification of endometriosis.Hum Reprod. 2017 Feb;32(2):315-324. doi: 10.1093/humrep/dew293. Epub 2016 Dec 5. Hum Reprod. 2017. PMID: 27920089
-
World Endometriosis Research Foundation Endometriosis Phenome and Biobanking Harmonization Project: II. Clinical and covariate phenotype data collection in endometriosis research.Fertil Steril. 2014 Nov;102(5):1223-32. doi: 10.1016/j.fertnstert.2014.07.1244. Epub 2014 Sep 22. Fertil Steril. 2014. PMID: 25256930 Free PMC article. Review.
-
EPHect - the Endometriosis Phenome (and Biobanking) Harmonisation Project - may be very helpful for clinicians and the women they are treating.F1000Res. 2017 Jan 5;6:14. doi: 10.12688/f1000research.9850.1. eCollection 2017. F1000Res. 2017. PMID: 28529694 Free PMC article. Review.
Cited by
-
The most impactful endometriosis symptom: An international, cross-sectional, two-round survey study.Acta Obstet Gynecol Scand. 2024 Sep;103(9):1736-1744. doi: 10.1111/aogs.14927. Epub 2024 Jul 23. Acta Obstet Gynecol Scand. 2024. PMID: 39041353 Free PMC article.
-
Endometriosis and adenomyosis research priorities in India and Sri Lanka: a call for regional collaboration.Lancet Reg Health Southeast Asia. 2025 Feb 14;34:100547. doi: 10.1016/j.lansea.2025.100547. eCollection 2025 Mar. Lancet Reg Health Southeast Asia. 2025. PMID: 40026836 Free PMC article. Review.
References
-
- ACOG Committee on Practice Bulletins - Gynecology 2000. ACOG practice bulletin. Medical management of endometriosis. Number 11, December 1999 (replaces Technical Bulletin Number 184, September 1993). Clinical management guidelines for obstetrician-gynecologists. International Journal of Gynaecology and Obstetrics 11183–196. (10.1016/s0020-7292(0100414-3) - DOI - PubMed
LinkOut - more resources
Full Text Sources

