Immunity and reproduction protective effects of Chitosan Oligosaccharides in Cyclophosphamide/Busulfan-induced premature ovarian failure model mice
- PMID: 37228612
- PMCID: PMC10203494
- DOI: 10.3389/fimmu.2023.1185921
Immunity and reproduction protective effects of Chitosan Oligosaccharides in Cyclophosphamide/Busulfan-induced premature ovarian failure model mice
Abstract
Introduction: Premature ovarian failure (POF) is a major cause of infertility among women of reproductive age. Unfortunately, there is no effective treatment available currently. Researchers have shown that immune disorders play a significant role in the development of POF. Moreover, growing evidence suggest that Chitosan Oligosaccharides (COS), which act as critical immunomodulators, may have a key role in preventing and treating a range of immune related reproductive diseases.
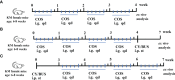
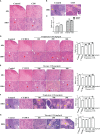
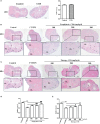
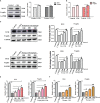
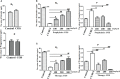
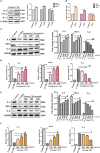
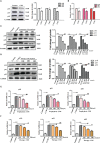
Methods: KM mice (6-8 weeks) received a single intraperitoneal injection of cyclophosphamide (CY, 120mg/kg) and busulfan (BUS, 30mg/kg) to establish POF model. After completing the COS pre-treatment or post-treatment procedures, peritoneal resident macrophages (PRMs) were collected for neutral erythrophagocytosis assay to detect phagocytic activity. The thymus, spleen and ovary tissues were collected and weighed to calculate the organ indexes. Hematoxylin-eosin (HE) staining was performed to observe the histopathologic structure of those organs. The serum levels of estrogen (E2) and progesterone (P) were measured via the enzyme-linked immunosorbent assay (ELISA). The expression levels of immune factors including interleukin 2 (IL-2), interleukin 4 (IL-4), and tumor necrosis factor α (TNF-α), as well as germ cell markers Mouse Vasa Homologue (MVH) and Fragilis in ovarian tissue, were analyzed by Western blotting and qRT-PCR. In addition, ovarian cell senescence via p53/p21/p16 signaling was also detected.
Results: The phagocytic function of PRMs and the structural integrity of thymus and spleen were preserved by COS treatment. The levels of certain immune factors in the ovaries of CY/BUS- induced POF mice were found to be altered, manifested as IL-2 and TNF-α experiencing a significant decline, and IL-4 presenting a notable increase. Both pre-treatment and post-treatment with COS were shown to be protective effects against the damage to ovarian structure caused by CY/BUS. Senescence-associated β-galactosidase (SA-β-Gal) staining results showed that COS prevents CY/BUS-induced ovarian cell senescence. Additionally, COS regulated estrogen and progesterone levels, enhanced follicular development, and blocked ovarian cellular p53/p21/p16 signaling which participating in cell senescence.
Conclusion: COS is a potent preventative and therapeutic medicine for premature ovarian failure by enhancing both the ovarian local and systemic immune response as well as inhibiting germ cell senescence.
Keywords: chitosan oligosaccharides; immunological function; ovarian function; premature ovarian failure; prophylaxis; therapy.
Copyright © 2023 Li, Ye, Su, Hu, Huang, Fu, Zhong, Du and Zheng.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials
Miscellaneous

