High‑dose aflibercept injection has striking effects on myopic choroidal neovascularization
- PMID: 37229317
- PMCID: PMC10203910
- DOI: 10.3892/etm.2023.12000
High‑dose aflibercept injection has striking effects on myopic choroidal neovascularization
Abstract
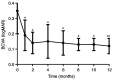
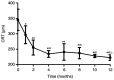
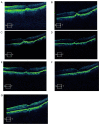
The aim of the present study was to evaluate the 1-year outcomes of a high-dose aflibercept injection [4 mg 2+ pro re nata (PRN) scheme] for individuals with myopic choroidal neovascularization (mCNV) through optical coherence tomography (OCT) follow-ups. A total of 16 consecutive patients (7 males and 9 females; sixteen eyes) with mCNV were enrolled in this retrospective study. The mean age was 30.5±3.35 years and mean spherical equivalent was -7.31±0.90 D. Subjects received 4 mg aflibercept intravitreal injection on the day of diagnosis and 35 days later. Further injections of aflibercept were required when the following were detected by OCT and fluorescein angiography: i) Decrease in best corrected visual acuity (BCVA); ii) aggravation of metamorphopsia; iii) macular oedema; iv) macular haemorrhage; v) increase in retinal thickness; and vi) leakage. Ophthalmic examination and OCT were performed at the baseline, as well as at 1, 2, 4, 6, 8, 10 and 12 months after the initial aflibercept injection. BCVA and central retinal thickness (CRT) were evaluated at each follow-up. The results showed that the vision of all subjects improved following the aflibercept intravitreal injection. The mean BCVA improved from 0.35±0.15 logarithm of the minimal angle of resolution (logMAR) at the baseline to 0.12±0.05 logMAR at final follow-up (P<0.05). A reduction in metamorphopsia was observed and the mean CRT was reduced from 345.38±34.69 µm of pre-treatment levels to 222.75±8.98 µm at the last postoperative visit (P<0.05). The mean number of injections in the present study was 2.13±0.5. Out of all patients, 13 received two injections and 3 subjects received three injections. The mean follow-up was 13.41±1.17 months. Based on the outcomes, it was found that an intravitreal injection of high-dose aflibercept (4 mg 2+PRN scheme) is effective for vision improvement and stabilization. In addition, it also significantly alleviated metamorphopsia and reduced the CRT in patients treated with mCNV. During the follow-up, the eyesight of the patients was stable.
Keywords: aflibercept; anti-vascular endothelial growth factor; choroidal neovascularization; optical coherence tomography; pathologic myopia.
Copyright: © Zhu et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures
Similar articles
-
Long-Term Outcomes of High-Dose Conbercept Treatment for Myopic Choroidal Neovascularization and Idiopathic Choroidal Neovascularization.Ophthalmic Res. 2023;66(1):636-644. doi: 10.1159/000529342. Epub 2023 Feb 6. Ophthalmic Res. 2023. PMID: 36746135
-
Intravitreal aflibercept versus bevacizumab for treatment of myopic choroidal neovascularization.Sci Rep. 2018 Sep 26;8(1):14389. doi: 10.1038/s41598-018-32761-z. Sci Rep. 2018. PMID: 30258077 Free PMC article.
-
Retinal vascular changes and aqueous humor cytokines changes after aflibercept intravitreal injection in treatment-naïve myopic choroidal neovascularization.Sci Rep. 2018 Oct 23;8(1):15631. doi: 10.1038/s41598-018-33926-6. Sci Rep. 2018. PMID: 30353107 Free PMC article. Clinical Trial.
-
Result of intravitreal aflibercept injection for myopic choroidal neovascularization.BMC Ophthalmol. 2021 Sep 22;21(1):342. doi: 10.1186/s12886-021-02088-x. BMC Ophthalmol. 2021. PMID: 34551746 Free PMC article.
-
HOW VITREOMACULAR INTERFACE MODIFIES THE EFFICACY OF ANTI-VEGF THERAPY FOR MYOPIC CHOROIDAL NEOVASCULARIZATION.Retina. 2018 Jan;38(1):84-90. doi: 10.1097/IAE.0000000000001500. Retina. 2018. PMID: 28098725
Cited by
-
Prognostic factors for intravitreal conbercept in the treatment of choroidal neovascularization secondary to pathological myopia.Int Ophthalmol. 2024 Jun 22;44(1):253. doi: 10.1007/s10792-024-03177-8. Int Ophthalmol. 2024. PMID: 38907787
References
LinkOut - more resources
Full Text Sources
Research Materials
