Phase I trial outcome of amnion cell therapy in patients with ischemic stroke (I-ACT)
- PMID: 37229431
- PMCID: PMC10204798
- DOI: 10.3389/fnins.2023.1153231
Phase I trial outcome of amnion cell therapy in patients with ischemic stroke (I-ACT)
Erratum in
-
Corrigendum: Phase I trial outcome of amnion cell therapy in patients with ischemic stroke (I-ACT).Front Neurosci. 2023 Jul 31;17:1237920. doi: 10.3389/fnins.2023.1237920. eCollection 2023. Front Neurosci. 2023. PMID: 37600008 Free PMC article.
Abstract
Background: We proposed a Phase I dose escalation trial to assess the safety of allogeneic human amniotic epithelial cells (hAECs) in stroke patients with a view to informing the design for a Phase II trial.
Methods: The design is based on 3 + 3 dose escalation design with additional components for measuring MR signal of efficacy as well as the effect of hAECs (2-8 × 106/kg, i.v.) on preventing immunosuppression after stroke.
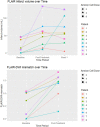
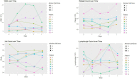
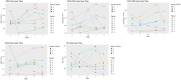
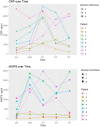
Results: Eight patients (six males) were recruited within 24 h of ischemic stroke onset and were infused with hAECs. We were able to increase the dose of hAECs to 8 × 106 cells/kg (2 × 106/kg, n = 3; 4 × 106/kg, n = 3; 8 × 106/kg, n = 2). The mean age is 68.0 ± 10.9 (mean ± SD). The frequencies of hypertension and hyperlipidemia were 87.5%, diabetes was 37.5%, atrial fibrillation was 50%, ischemic heart disease was 37.5% and ever-smoker was 25%. Overall, baseline NIHSS was 7.5 ± 3.1, 7.8 ± 7.2 at 24 h, and 4.9 ± 5.4 at 1 week (n = 8). The modified Rankin scale at 90 days was 2.1 ± 1.2. Supplemental oxygen was given in five patients during hAEC infusion. Using pre-defined criteria, two serious adverse events occurred. One patient developed recurrent stroke and another developed pulmonary embolism whilst in rehabilitation. For the last four patients, infusion of hAECs was split across separate infusions on subsequent days to reduce the risk for fluid overload.
Conclusion: Our Phase I trial demonstrates that a maximal dose of 2 × 106/kg hAECs given intravenously each day over 2 days (a total of 4 × 106/kg) is safe and optimal for use in a Phase II trial.
Clinical trial registration: ClinicalTrials.gov, identifier ACTRN12618000076279P.
Keywords: allogeneic; clinical trial; ischemic stroke; phase I; stem cell.
Copyright © 2023 Phan, Lim, Chan, McDonald, Gan, Zhang, Barreto Arce, Vuong, Thirugnanachandran, Clissold, Ly, Singhal, Hervet, Kim, Drummond, Wallace, Ma and Sobey.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
LinkOut - more resources
Full Text Sources
Other Literature Sources

